
- Healthcare Services
- Life Sciences Electronic Batch Records Market
Life Sciences Electronic Batch Records Market Size, Share, and Growth Forecast, 2026 - 2033
Life Sciences Electronic Batch Records Market by Product Type (Cloud-based EBR Solutions, On-premise EBR Solutions), Functionality (Data Management, Reporting and Analytics, Others), End-user, and Regional Analysis for 2026 - 2033
Life Sciences Electronic Batch Records Market Size and Trends Analysis
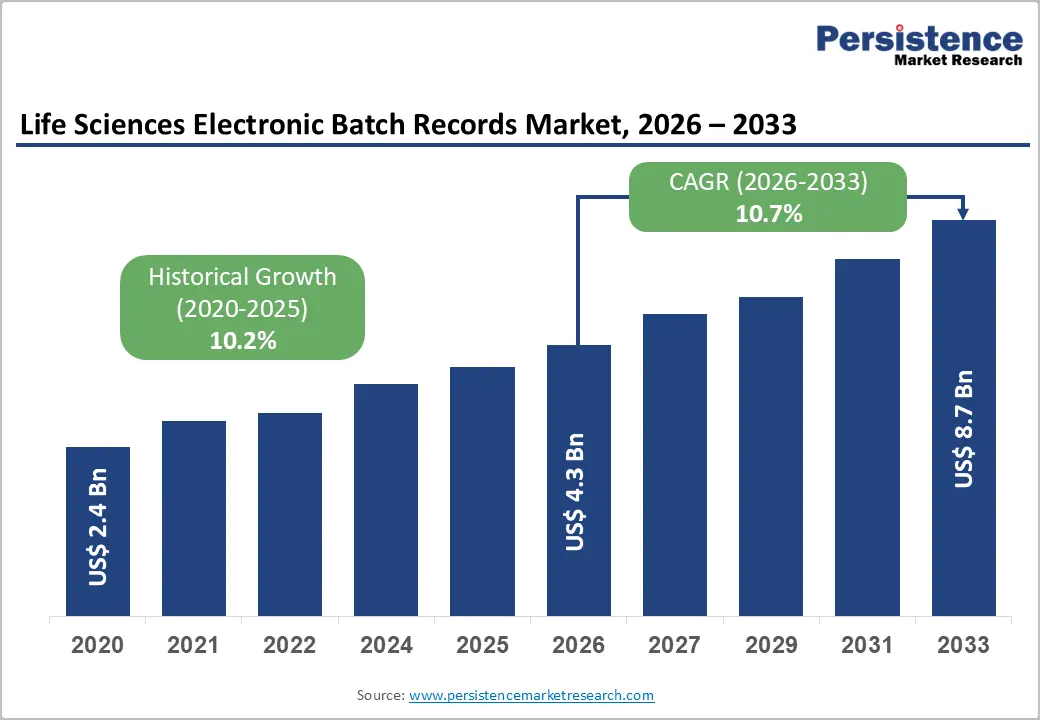
The global life sciences electronic batch records market size is likely to be valued at US$4.3 billion in 2026, and is expected to reach US$8.7 billion by 2033, growing at a CAGR of 10.7% during the forecast period from 2026 to 2033, driven by stringent regulatory requirements, the need for paperless manufacturing, and the push for operational efficiency in life sciences production. Increasing adoption of digital batch documentation to ensure compliance, traceability, and real-time visibility remains a major driver of the life sciences electronic batch records market growth.
Key Industry Highlights:
- Leading Region: North America, anticipated to account for a 39% market share in 2026, driven by advanced pharmaceutical manufacturing and strict regulatory standards in the U.S.
- Fastest-growing Region: Asia Pacific, fueled by expanding biopharma manufacturing, rising GMP compliance focus, and digital transformation in China and India.
- Dominant Product Type: On-premises EBR solutions are set to dominate in 2026, accounting for 60% of the market share, driven by large manufacturers prioritizing data control, security, and deep system integration in regulated environments.
- Leading Functionality: Compliance and quality control will dominate 50% of the EBR market in 2026, driven by automation, audit trails, and support for regulatory compliance and product quality.
| Key Insights | Details |
|---|---|
|
Life Sciences Electronic Batch Records Market Size (2026E) |
US$4.3 Bn |
|
Market Value Forecast (2033F) |
US$8.7 Bn |
|
Projected Growth CAGR (2026-2033) |
10.7% |
|
Historical Market Growth (2020-2025) |
10.2% |
DRO Analysis
Driver - Shift from Paper-Based to Digital Documentation
In life sciences manufacturing, traditional paper-based batch records have long been the norm, but they are increasingly viewed as inefficient and prone to errors. Manual documentation requires extensive time and labor, increases the risk of mistakes, and complicates the retrieval of historical data during audits or regulatory inspections. Electronic batch records (EBR) address these challenges by digitizing the entire documentation process, offering automated workflows that ensure accuracy, consistency, and traceability.
Digital systems streamline the creation, execution, and review of batch records, reducing manual intervention and minimizing human errors. They allow real-time data entry and validation, making it easier for staff to identify discrepancies immediately rather than discovering them during later audits. The transition also enhances collaboration across departments, as authorized personnel can access records simultaneously from different locations, improving efficiency in production and quality management. Electronic systems support regulatory compliance by providing audit trails, secure access, and standardized documentation formats, which are difficult to achieve with paper records.
Enhanced Data Accuracy & Quality Management
Maintaining precise and consistent data is crucial in life sciences manufacturing, where even minor errors can impact product quality, regulatory compliance, and patient safety. Traditional paper-based systems are susceptible to mistakes such as misentries, lost records, and incomplete documentation, which can delay batch approvals or trigger compliance issues. Electronic batch record (EBR) systems address these challenges by providing automated data capture, validation, and storage, ensuring that all entries are accurate, complete, and traceable.
By digitizing production and quality workflows, EBR systems reduce manual intervention and enforce standard operating procedures consistently. Real-time checks and validation rules help catch deviations or errors immediately, preventing flawed data from progressing through the manufacturing process. This not only minimizes rework and production delays but also strengthens the reliability of quality control processes. Electronic records facilitate seamless data integration across systems, enabling cross-functional teams to monitor trends, perform audits, and generate reports quickly. Centralized and secure data storage enhances traceability, making it easier to respond to regulatory inspections or quality investigations.
Restraint - High Initial Implementation Cost
Implementing electronic batch record (EBR) systems in life sciences manufacturing requires significant upfront investment, which can be a major hurdle for many organizations. The costs involve purchasing or licensing specialized software, upgrading or installing compatible hardware, and integrating the system with existing platforms such as MES, ERP, or LIMS. Extensive validation, testing, and compliance procedures must be conducted to meet regulatory standards, further increasing the initial expenditure.
Training staff to use the new system effectively adds another layer of cost, as employees must become proficient in digital workflows and quality protocols. Smaller organizations or those with limited budgets may find these requirements prohibitive, delaying adoption or limiting system functionality. Despite long-term efficiency gains, the substantial upfront financial commitment and resource allocation required can slow decision-making and implementation, making organizations weigh the short-term costs against potential long-term benefits carefully.
Workforce Resistance and Training Needs
Adopting electronic batch record (EBR) systems often requires a cultural and operational shift within life sciences organizations. Employees accustomed to paper-based processes may be reluctant to change, perceiving digital systems as complex or threatening to established routines. This resistance can slow system adoption, reduce efficiency during the transition, and impact overall productivity if not managed effectively.
To address these challenges, comprehensive training programs are essential. Staff must learn how to navigate the software, enter and validate data accurately, and follow new digital workflows that align with regulatory requirements. Training also helps employees understand the benefits of the system, such as reduced errors, faster approvals, and improved data traceability, which can increase buy-in and reduce resistance. Ongoing support and hands-on guidance are critical during the early stages of implementation.
Opportunity - Rapid Cloud-Based EBR Adoption
Life sciences companies are increasingly moving their electronic batch record (EBR) systems to cloud-based platforms, driven by the need for scalability, flexibility, and cost efficiency. Cloud solutions eliminate the need for extensive on-site IT infrastructure, reducing capital expenditure while providing access to advanced software capabilities. This allows organizations of all sizes, including small and medium enterprises, to implement sophisticated EBR systems without heavy upfront investments.
Cloud-based EBR systems enable real-time access to batch records from multiple sites, supporting decentralized operations and remote monitoring. This accessibility improves collaboration between production, quality, and regulatory teams, allowing them to review, approve, and analyze records more efficiently. Cloud platforms often offer automatic updates and maintenance, ensuring that the system remains compliant with evolving regulatory requirements without significant internal resource allocation. Security and data integrity are enhanced through cloud providers’ built-in encryption, backup, and disaster recovery mechanisms. Cloud deployment supports integration with other digital manufacturing systems, including MES, ERP, and quality management platforms, enabling seamless data flow and process automation.
Digital Transformation in Pharma & Biotech
Pharmaceutical and biotechnology companies are increasingly embracing digital transformation to improve efficiency, compliance, and innovation across the entire product lifecycle. By replacing manual, paper-based processes with automated, data-driven systems, organizations can streamline operations from research and development to manufacturing, quality control, and regulatory reporting. Electronic batch records (EBRs), laboratory information management systems (LIMS), and digital quality management platforms are key components that enable this transformation.
Digital tools allow real-time tracking of production and quality data, improving transparency and traceability while reducing errors associated with manual documentation. Integration of these systems with enterprise platforms such as ERP and manufacturing execution systems (MES) enhances coordination across departments, enabling faster decision-making and more efficient resource allocation. Digital transformation supports advanced analytics and predictive insights, allowing organizations to optimize processes, identify potential risks, and accelerate product development timelines. For biotech firms working on complex biologics or personalized therapies, digitalization ensures precise control over manufacturing parameters and rigorous compliance with regulatory standards.
Category-wise Analysis
Product Type Insights
On-premise EBR solutions are projected to dominate, holding 60% of the market share in 2026, driven by large pharmaceutical and biotech manufacturers that prioritize direct control over sensitive production data, stringent data security policies, and deep integration with existing internal IT systems such as MES, ERP, and quality platforms. On-premise systems also appeal to organizations operating in highly regulated environments where internal governance, offline access, and customization are critical to meeting compliance obligations and managing complex batch processes. The Werum PAS-X MES platform, featuring integrated EBR functionality, has been adopted by several pharmaceutical manufacturers, including Thailand’s Government Pharmaceutical Organization. Using their own infrastructure, they achieved paperless operations, real-time exception review, and faster batch release while ensuring strict GMP compliance.
Cloud-based EBR solutions are likely to be the fastest-growing, due to their flexibility, scalability, and ease of deployment. Companies can access batch records remotely, enabling real-time monitoring, approvals, and collaboration across multiple sites without being tied to on-premise infrastructure. These solutions reduce upfront costs by eliminating the need for extensive hardware and IT maintenance, making them especially attractive to small and medium-sized enterprises. bioMérieux, a global life sciences firm, moved from paper-based batch records to a digital EBR system using MasterControl’s cloud-enabled Manufacturing Excellence (Mx) software. This implementation allowed the company to automate data capture, enhance data integrity by connecting process equipment directly to the system, and minimize manual entry errors previously common with paper documentation.
Functionality Insights
The compliance and quality control segment is projected to dominate, accounting 50% of the market share in 2026, fueled by the essential role EBR systems play in helping pharmaceutical and biotechnology manufacturers meet stringent regulatory standards and maintain consistent product quality. By automating documentation, enforcing procedural controls, and providing comprehensive audit trails, EBR solutions reduce manual errors and ensure accurate, traceable records that are critical during regulatory inspections and audits. Pfizer utilizes automated EBR systems to digitize batch records, ensuring accurate, tamper-evident documentation and real-time audit trails. This supports GMP compliance and regulatory standards such as FDA 21 CFR Part 11, improves batch review efficiency, and strengthens overall quality control in manufacturing operations.
The reporting and analytics segment is expected to be the fastest-growing functionality, propelled by the increasing demand for real-time insights into production and quality processes. Advanced analytics enable manufacturers to monitor batch performance, identify deviations, and optimize workflows promptly, reducing errors and improving operational efficiency. By leveraging automated data collection and visualization tools, organizations can make data-driven decisions, streamline compliance reporting, and enhance overall process transparency. Alkaloid AD Skopje implemented Accevo Systems’ EBR solution to access real-time batch reports and production analytics, enhancing visibility, quality oversight, and operational efficiency while enabling faster, data-driven decision-making.

Regional Insights
North America Life Sciences Electronic Batch Records Market Trends
North America is projected to dominate, capturing 39% of revenue in 2026, powered by the region’s advanced regulatory landscape and high concentration of pharmaceutical and biotechnology companies. Manufacturers are increasingly adopting EBR systems to ensure regulatory compliance, streamline operations, and improve product quality. Stringent standards set by agencies such as the FDA, including 21 CFR Part 11, have made electronic record-keeping not just beneficial but essential, prompting companies to digitize their batch documentation processes.
Technological adoption is another key trend, with organizations integrating EBR systems with Manufacturing Execution Systems (MES), Enterprise Resource Planning (ERP), and Laboratory Information Management Systems (LIMS). This connectivity enables seamless data flow, real-time monitoring, and process automation, reducing manual errors and accelerating batch approvals. Cloud-based deployments are growing rapidly, driven by the need for remote access, scalability, and lower upfront IT costs, while on-premise solutions continue to dominate large enterprises requiring full control over sensitive data. North American companies are leveraging analytics and reporting functionalities in EBR systems to gain actionable insights into production efficiency, deviations, and quality trends.
Europe Life Sciences Electronic Batch Records Market Trends
Europe market is evolving as pharmaceutical and biotech manufacturers increasingly pursue digital transformation to enhance compliance and operational efficiency. European regulatory requirements, such as those enforced by the EMA and national agencies, emphasize data integrity and traceability, prompting companies to replace paper-based batch documentation with digital solutions that provide secure audit trails and consistent quality records. This regulatory focus is encouraging wider adoption of EBR systems across the region.
Manufacturers are also integrating EBR platforms with other enterprise systems such as Manufacturing Execution Systems (MES), ERP, and quality management software to create interconnected digital ecosystems. This integration improves real-time visibility into production processes, reduces manual errors, and supports faster decision-making. While large multinational companies tend to favor on-premises solutions due to data control and internal infrastructure preferences, cloud-based EBR deployments are gaining momentum, especially among mid-sized firms and contract manufacturing organizations seeking lower upfront costs, scalability, and remote access.
Asia Pacific Life Sciences Electronic Batch Records Market Trends
Asia Pacific is projected to be the fastest-growing region, driven by increasing adoption of digital record-keeping among pharmaceutical and biotechnology companies to improve product quality, strengthen compliance, and enhance operational efficiency. Historically reliant on paper-based documentation, many companies across countries such as India, China, Japan, and South Korea are now investing in EBR systems to keep pace with global regulatory expectations and to support export-oriented production.
Regulatory agencies in the region are placing greater emphasis on data integrity, traceability, and audit readiness, which is encouraging life sciences firms to modernize their documentation processes.
In the Asia Pacific market, cloud-based electronic batch record (EBR) solutions are gaining traction, especially among small and medium-sized manufacturers that often lack extensive in-house IT capabilities. Cloud deployment allows these firms to adopt advanced systems more rapidly and with lower upfront investment, while also enabling remote access and easy scalability across multiple locations.
In contrast, larger enterprises tend to favor on-premise solutions due to their preference for greater data control and the need to integrate with existing manufacturing execution systems (MES) and enterprise resource planning (ERP) platforms. At the same time, there is a growing emphasis on integrating EBR systems with broader digital ecosystems, including ERP, MES, and quality management software, to enable real-time monitoring, minimize manual errors, and improve overall operational efficiency.
Organizations are also increasingly utilizing reporting and analytics capabilities within EBR platforms to extract actionable insights from production data. This helps identify trends, optimize batch performance, and support continuous improvement efforts.

Competitive Landscape
The global life sciences electronic batch records (EBR) market is highly competitive, driven by the need for digital solutions that ensure regulatory compliance, scalability, and seamless integration with existing manufacturing and quality systems. Technology providers differentiate themselves through advanced cloud capabilities, intuitive user interfaces, and comprehensive validation support tailored for life sciences operations. Leading companies such as Rockwell Automation, Accelrys, Siemens, ABB, Emerson Electric, Honeywell International, and Schneider Electric are actively investing in next-generation EBR platforms to meet the evolving demands of pharmaceutical and biotechnology manufacturers.
Key strategies in this competitive landscape include developing fully validated cloud-based solutions that enable remote access, multi-site operations, and real-time monitoring. Companies are also strengthening partnerships with pharma and biotech firms, integrating robust cybersecurity and compliance features, and expanding into high-growth Asian markets to capitalize on the region’s increasing adoption of digital manufacturing.
Key Industry Developments:
- In May 2025, MasterControl, a leading provider of quality, manufacturing, and asset management software solutions, announced the beta release of its Master Template Generator at the Master's Conference in Berlin. The new AI-powered tool enabled manufacturers using MasterControl's Manufacturing Excellence (Mx) Production Records module to optimize batch records more efficiently. It streamlined the transition from paper-based documentation to digital manufacturing execution, helping companies reduce manual effort, enhance accuracy, and accelerate compliance processes across production workflows.
Companies Covered in Life Sciences Electronic Batch Records Market
- Rockwell Automation
- Accelrys
- Siemens
- ABB
- Emerson Electric
- Honeywell international
- Schneider Electric
Frequently Asked Questions
The global life sciences electronic batch records market is projected to reach US$4.3 billion in 2026.
In life sciences manufacturing, electronic batch records (EBR) replace inefficient, error-prone paper records with automated, accurate, and traceable digital workflows.
The life sciences electronic batch records market is poised to witness a CAGR of 10.7% from 2026 to 2033.
Life sciences companies are adopting cloud-based EBR systems for scalable, flexible, and cost-efficient digital batch management without heavy IT infrastructure.
Key players in the life sciences electronic batch records market include Rockwell Automation, Siemens, Emerson Electric, Honeywell International, and Schneider Electric.







