
- Biotechnology
- TCR-Based Antibody Market
TCR-Based Antibody Market Size, Share, and Growth Forecast, 2026 - 2033
TCR-Based Antibody Market by Target Antigen (NY-ESO-1, p53, WT-1, EBV), Indication (Bladder Cancer, Multiple Myeloma, Others), End-user (Hospitals, Specialized Clinics, Pharma and Biotech Research Laboratories, Others), and Regional Analysis for 2026 - 2033
TCR-Based Antibody Market Size and Trends Analysis
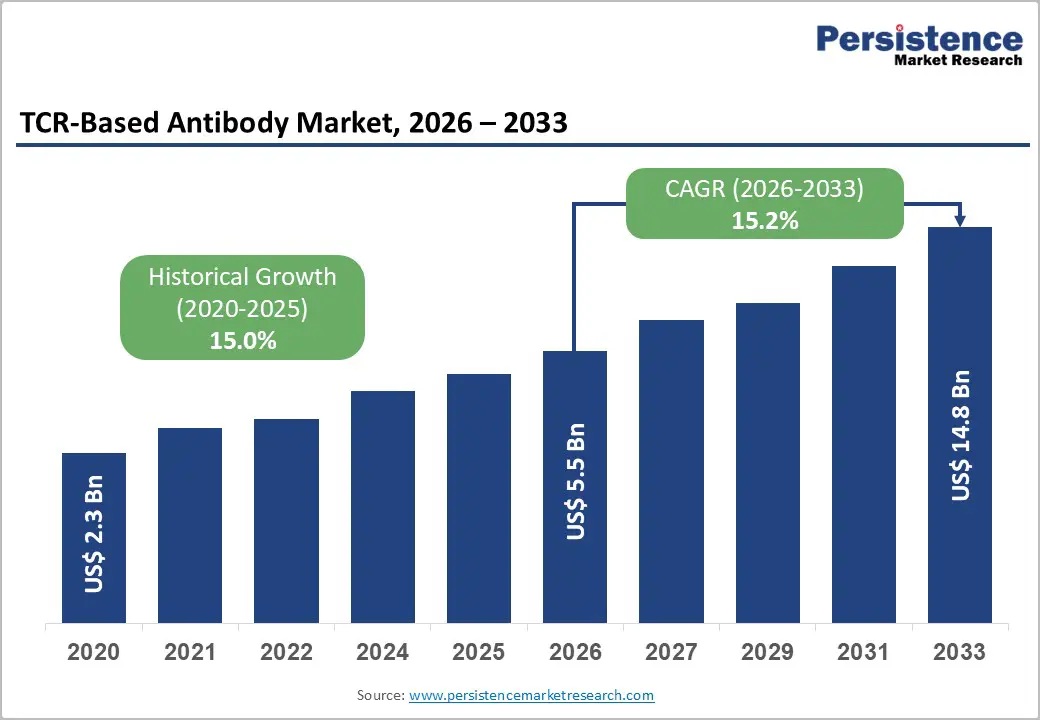
The global TCR-based antibody market size is likely to be valued at US$5.5 billion in 2026 and is expected to reach US$14.8 billion by 2033, growing at a CAGR of 15.2% during the forecast period from 2026 to 2033, driven by increasing adoption of precision immunotherapies, rising global cancer incidence, and advancements in TCR engineering and affinity maturation technologies.
This expanded targeting potential enables these therapies to target a wide range of cancer-specific antigens, including NY-ESO-1, p53, WT-1, and EBV-derived epitopes, making them particularly valuable for treating both solid tumors and hematologic malignancies characterized by heterogeneous antigen profiles. The growth of this market aligns with trends in cancer.
According to the World Health Organization’s 2024 estimates, approximately 20 million new cancer cases were diagnosed worldwide in 2022, reflecting a sustained high incidence that drives the need for novel and precise treatment modalities. Technological advancements in TCR engineering, affinity maturation, bispecific and multispecific formats, and high-throughput screening have significantly enhanced immune engagement and therapeutic specificity.
Key Industry Highlights:
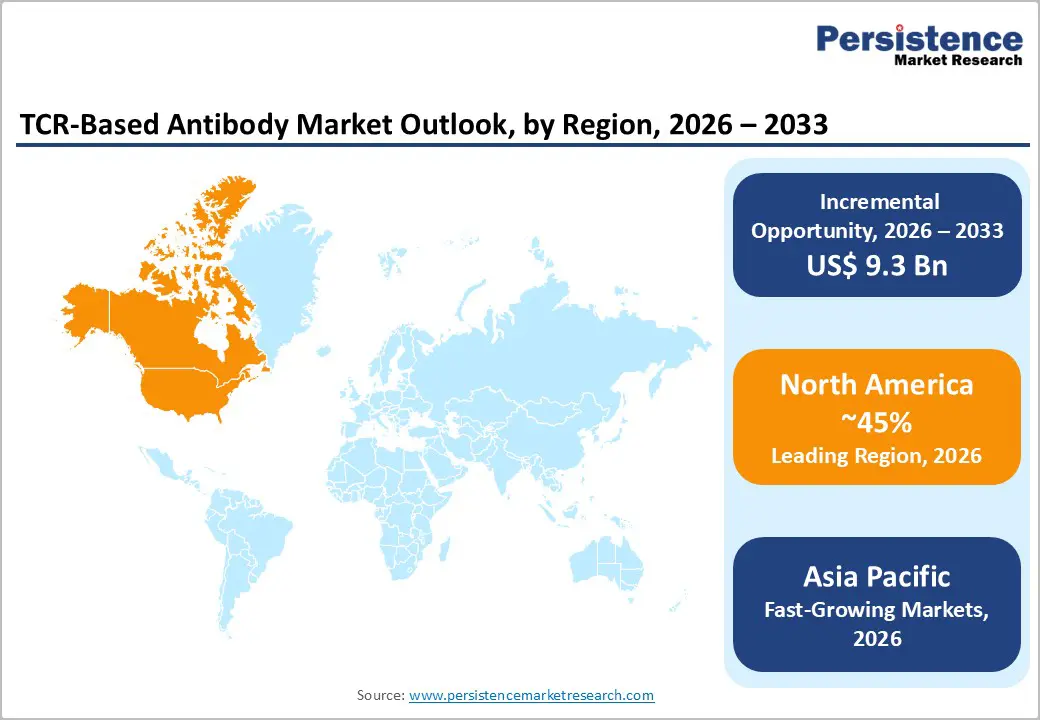
- Leading Region: North America is anticipated to be the leading region, accounting for 45% market share in 2026, driven by strong innovation, regulatory support, a dense clinical trial infrastructure, and high adoption of advanced immunotherapies.
- Fastest-growing Region: Asia Pacific is likely to be the fastest-growing region, supported by rapid adoption, growing biotech investment, regulatory support, and large patient populations.
- Leading Target Antigen: NY-ESO-1 is projected to be the leading target antigen in 2026, accounting for 40% of revenue share, driven by its high cancer-testis antigen specificity and extensive clinical validation across multiple tumor types.
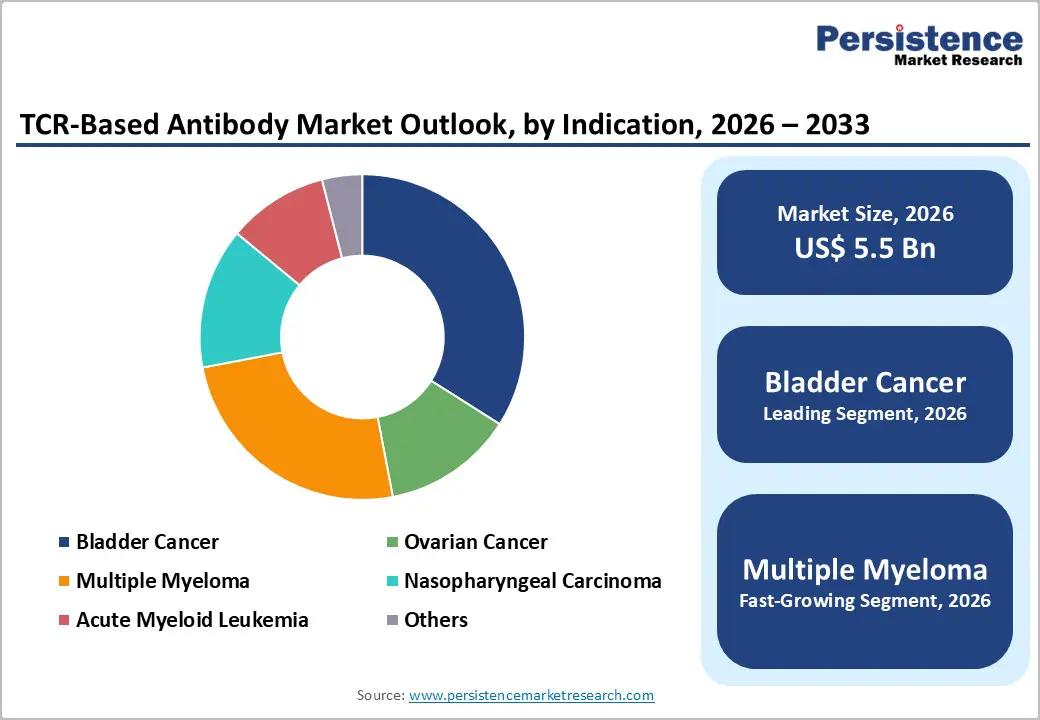
- Leading Indication: Bladder cancer is anticipated to be the leading indication, accounting for over 43% of the revenue share in 2026, supported by high antigen expression and limited durable responses to existing therapies.
| Key Insights | Details |
|---|---|
| TCR-Based Antibody Market Size (2026E) | US$5.5 Bn |
| Market Value Forecast (2033F) | US$14.8 Bn |
| Projected Growth (CAGR 2026 to 2033) | 15.2% |
| Historical Market Growth (CAGR 2020 to 2025) | 15.0% |
DRO Analysis
Driver - Advancements in TCR Engineering and Target Identification
TCR-based antibody development has greatly benefited from advances in T-cell receptor engineering, including affinity maturation, bispecific formats, and high-throughput screening. These advances enable precise recognition of intracellular peptide-MHC complexes, expanding the spectrum of targetable cancer antigens beyond conventional surface markers. Sophisticated bioinformatics and structural modeling allow rapid identification of tumor-specific epitopes, including NY-ESO-1, p53, and WT-1, improving therapeutic specificity and efficacy.
Continued refinement of TCR design supports more potent and selective immune engagement, enabling combination with other modalities, such as checkpoint inhibitors or CAR-T therapies. Enhanced target identification minimizes off-tumor effects, enabling safer dosing regimens and broader patient applicability. Advancements in peptide-MHC binding prediction and epitope mapping have expanded pipelines across multiple cancer types, from sarcoma to hematologic malignancies.
Rising Cancer Burden and Demand for Precision Oncology
An increase in cancer incidence has intensified demand for therapies capable of addressing heterogeneous tumor profiles. Traditional treatments often fail to deliver durable responses in solid tumors or relapsed hematologic malignancies, creating opportunities for TCR-based antibodies. These therapies target intracellular antigens, offering precision treatment options tailored to individual tumor biology. Rising awareness of personalized medicine and patient-specific therapies has driven adoption, particularly in regions with advanced healthcare infrastructure.
Expanding cancer prevalence, particularly in aging populations, fuels continuous clinical demand. Precision oncology frameworks and biomarker-driven treatment protocols increase acceptance of TCR-based approaches in hospitals, specialized clinics, and research laboratories. Combining TCR-based antibodies with existing therapies enhances clinical outcomes while addressing unmet needs in aggressive or treatment-resistant cancers.
Restraint - Technical Challenges in Manufacturing and Scalability
TCR-based antibodies involve complex protein engineering, requiring precise folding, stability, and affinity optimization. Manufacturing these biologics at scale presents significant challenges, including maintaining batch-to-batch consistency and high purity standards. Specialized facilities, stringent quality control, and advanced bioprocessing capabilities are required, increasing production costs. Scaling clinical-grade production from preclinical studies to large commercial batches is technically demanding and limits accessibility.
Production complexity also affects cost-effectiveness, delaying commercialization in emerging regions. Regulatory oversight mandates rigorous validation of manufacturing processes, adding time and expense. Integration of automated platforms and bioreactors helps, but requires significant investment. Scalability constraints limit the ability to conduct multi-center clinical trials and achieve broad commercial distribution, creating barriers to widespread adoption.
Potential Safety Risks and Off-Target Effects
TCR-based antibodies, by targeting intracellular antigens via peptide-MHC complexes, carry inherent risks of off-target recognition and immune-related adverse events. Cross-reactivity with non-tumor tissues can trigger cytotoxicity, potentially causing severe organ-specific damage. Safety concerns limit dosing strategies and may result in regulatory delays or additional clinical monitoring requirements. Predicting off-target effects remains challenging due to complex immune interactions, requiring advanced preclinical modeling and extensive safety studies.
Immune-related toxicities, including cytokine release syndrome or autoimmunity, necessitate careful trial design and patient selection. These risks influence regulatory decisions, often requiring additional trials to demonstrate safety, thereby slowing development timelines. Risk mitigation strategies, such as affinity tuning and dual-targeting designs, are increasingly applied but add complexity to engineering and production. Managing potential adverse events is vital for clinician confidence, patient acceptance, and long-term market sustainability, making safety optimization a key challenge for TCR-based antibody commercialization.
Opportunity - Technological Convergence with Gene Editing and Combination Therapies
Integration of TCR-based antibodies with gene-editing platforms, such as CRISPR/Cas9, offers opportunities to enhance specificity and reduce immunogenicity. Combination therapies with checkpoint inhibitors, CAR-T cells, or other immunomodulators can increase efficacy, particularly in resistant tumors. These approaches enable synergistic immune activation while expanding the scope of targetable antigens. Technological convergence facilitates the development of next-generation TCR constructs with improved affinity, multi-epitope recognition, and safety profiles.
Emerging platforms enable the customization of patient-specific TCR therapies, creating opportunities for precision treatment. Integration with advanced delivery systems and gene-modification techniques supports more durable responses. By combining TCR-based antibodies with other immunotherapies, developers can address tumor heterogeneity and improve overall outcomes. Investment in convergent technologies strengthens competitive positioning and opens new therapeutic avenues in oncology.
Unmet Needs in Solid Tumors and Personalized Approaches
Solid tumors remain a major area of unmet medical need due to heterogeneous antigen expression and limited efficacy of conventional therapies. TCR-based antibodies provide access to intracellular tumor antigens, enabling treatment of previously intractable cancers. Personalized medicine approaches, guided by tumor-specific antigen profiling, allow clinicians to tailor therapies to individual patients, improving response rates. The ability to target rare or tumor-restricted antigens like NY-ESO-1 and EBV-derived epitopes positions TCR-based therapies as highly differentiated options in oncology.
Advances in diagnostic tools and molecular profiling enhance patient stratification, ensuring optimal therapeutic outcomes. Opportunities exist across multiple solid tumors, including ovarian cancer, bladder cancer, and nasopharyngeal carcinoma, where conventional treatments show limited durability. Regional prevalence data, combined with personalized antigen targeting, support expansion in both developed and emerging markets. Addressing these high-unmet-need areas strengthens clinical relevance, encourages investment, and solidifies the role of TCR-based antibodies in next-generation cancer treatment paradigms.
Category-wise Analysis
Target Antigen Insights
NY-ESO-1 is expected to lead the TCR-based antibody market, accounting for approximately 40% of revenue in 2026, driven by a strong presence in TCR-based antibody development. Its dominance is attributed to its high cancer-testis antigen specificity, which is expressed in tumor tissues but largely absent in normal adult tissues, making it an ideal candidate for targeted immunotherapy. For example, patients with advanced synovial sarcoma treated with NY-ESO-1-targeted TCR therapies have shown significant tumor regression, underscoring the clinical potential and investor confidence in this antigen.
WT-1 is likely to represent the fastest-growing segment, supported by its prevalence in common cancers and ongoing engineering innovations. These antigens are implicated in a broad spectrum of malignancies, with p53 mutations occurring in over half of all tumors. Advances in peptide-MHC binding prediction, affinity maturation, and combination strategies allow developers to mitigate cross-reactivity and enhance specificity. For example, WT-1-targeted TCR therapies have demonstrated encouraging activity in acute myeloid leukemia, highlighting their potential in hematologic malignancies.
Indication Insights
Bladder cancer is projected to lead the market, capturing around 43% of the revenue share in 2026, supported by high expression of relevant intracellular antigens and the limited efficacy of existing therapies in advanced or muscle-invasive disease. For example, TCR-based therapies targeting NY-ESO-1 have shown meaningful clinical responses in patients with advanced bladder cancer, demonstrating their capacity to address unmet clinical needs. The established clinical pipeline, regulatory support, and robust patient stratification practices enhance adoption across hospitals and specialized oncology clinics.
Multiple myeloma is likely to be the fastest-growing indication, driven by high unmet clinical needs and the ability of TCR-based antibodies to access intracellular targets. For example, NY-ESO-1-targeted TCR therapies have demonstrated promising results in relapsed/refractory multiple myeloma, highlighting their potential to improve patient outcomes where conventional treatments fail. The rising incidence, biomarker-driven patient selection, and maturation of clinical pipelines are driving the accelerated growth of these segments.
Regional Insights
North America TCR-Based Antibody Market Trends
North America is anticipated to be the leading region, accounting for a market share of 45% in 2026, driven by the U.S., which benefits from a robust biotechnology ecosystem, extensive clinical trial infrastructure, and supportive regulatory frameworks such as the FDA’s RMAT and Breakthrough Therapy designations. The high adoption of advanced immunotherapies in hospitals and specialized clinics reflects the growing acceptance of precision oncology approaches. Companies such as Adaptimmune Therapeutics PLC are actively advancing NY-ESO-1-targeted TCR therapies, demonstrating strong clinical efficacy in sarcoma and multiple myeloma.
The region’s market trends emphasize combination therapies, personalized approaches, and next-generation TCR constructs. Rising cancer incidence in aging populations fuels demand, while regulatory frameworks ensure safety and efficacy, enabling faster approvals. Adoption is also supported by favorable reimbursement policies, patient access programs, and increasing clinical trial enrollment. North America continues to serve as a hub of innovation, with leading biotech companies investing heavily in engineering TCRs targeting intracellular antigens such as p53, WT-1, and EBV, further strengthening the region’s leadership position in the TCR-based antibody market.
Europe TCR-Based Antibody Market Trends
Europe is likely to be a significant market for TCR-based antibodies, driven by a strong clinical research ecosystem and regulatory support from bodies such as the European Medicines Agency (EMA). The region hosts a growing number of biotechnology firms advancing TCR-focused therapies, reflecting a broader shift toward precision oncology. For example, T-Therapeutics, a UK-based biotech that recently raised significant Series-A funding to develop next-generation TCR therapeutics for cancer, indicates strong investor confidence in the European TCR landscape and its translational potential.
Collaborative efforts between European biotechs and pharmaceutical partners enhance development opportunities. The trend toward personalized immunotherapies, combined with advancements in TCR discovery platforms and biomarker-driven patient selection, strengthens the region’s ability to compete on the stage. Continued progress in clinical studies, coupled with supportive regulatory frameworks, drives the adoption of TCR-based antibodies across hospitals, specialized clinics, and research centers throughout Europe.
Asia Pacific TCR-Based Antibody Market Trends
The Asia Pacific region is likely to be the fastest-growing, driven by rising healthcare investments, supportive regulatory reforms, and expanding clinical research across China and Southeast Asia. Large patient populations and improving healthcare infrastructure enhance the region’s attractiveness for both domestic biotech firms and multinational companies seeking regional expansion. For example, Biosyngen Pte Ltd, a Singapore-based biotech advancing its BRG01 TCR therapy for solid tumors and EBV-positive malignancies, is currently in Phase I/II trials with regulatory approvals in the U.S. and China.
Trends in Asia Pacific emphasize strategic collaborations, technology transfer agreements, and growing public-private partnerships that strengthen R&D and manufacturing capacities. Local biopharma ecosystems are evolving rapidly, supported by faster IND approvals and increasing participation in clinical programs. Countries such as China are aligning their regulations with international standards, facilitating smoother pathways for novel biologics and personalized cancer therapies.
Competitive Landscape
The global TCR-based antibody market exhibits a moderately fragmented structure, driven by continuous innovation from both established pharmaceutical giants and specialized biotech firms that are expanding pipelines and advancing clinical research. Market momentum is shaped by robust R&D activities, strategic collaborations, and alliances that enable firms to refine TCR engineering, expand antigen targets, and accelerate late-stage clinical programs.
With key leaders including Adaptimmune Therapeutics PLC, Immunocore, Lion TCR, GlaxoSmithKline, and Merck & Co., the competitive space reflects a blend of deep scientific expertise and commercial scale that shapes therapeutic adoption and investment priorities. These companies are actively involved in advancing therapies targeting NY-ESO-1, WT-1, p53, and other intracellular antigens, moving candidates through Phase I/II programs and forming co-development partnerships to broaden geographical reach and share risk.
Key Industry Developments:
- In March 2026, Crossbow Therapeutics raised US$77 million in Series-B financing to advance its TCR-mimetic antibody therapy programs. The funding round, co-led by Taiho Ventures and Arkin Bio Capital with participation from investors including Pfizer Ventures and Eli-Lilly and Company, will support the completion of the Phase-1 CROSSCHECK-001 clinical trial for its lead candidate CBX-250 and accelerate development of additional T-Bolt™ immunotherapy candidates targeting peptide-HLA complexes across a broad range of cancers.
- In July 2025, Crossbow Therapeutics nominated its second TCR-mimetic development candidate, CBX-663, expanding its immuno-oncology pipeline. The bispecific T-cell engager targets telomerase reverse transcriptase (TERT) peptide-HLA complexes, which are widely expressed across many solid tumors and hematologic malignancies.
Companies Covered in TCR-Based Antibody Market
- Lion TCR
- GlaxoSmithKline
- Adaptimmune Therapeutics PLC
- Celgene Corporation
- Immunocore
- Kuur Therapeutics Limited
- Kite Pharma
- Takara Bio Inc.
- Ziopharm Oncology Inc.
- Merck & Co. Inc.
Frequently Asked Questions
The global TCR-based antibody market is projected to reach US$5.5 billion in 2026.
The TCR-based antibody market is driven by the growing demand for precision immunotherapies targeting intracellular tumor antigens inaccessible to conventional antibodies.
The TCR-based antibody market is expected to grow at a CAGR of 15.2% from 2026 to 2033.
Key market opportunities lie in developing therapies for solid tumors and personalized cancer treatments using advanced TCR-based platforms.
Lion TCR, GlaxoSmithKline, Adaptimmune Therapeutics PLC, Celgene Corporation, and Immunocore are the leading players.







