- Executive Summary
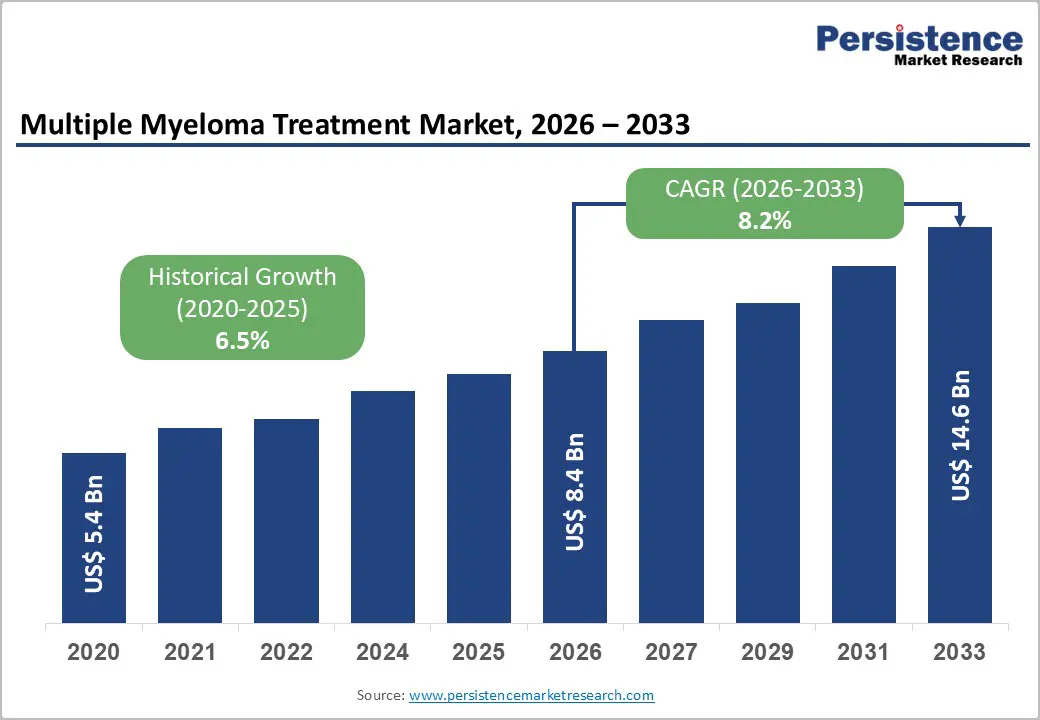
- Global Multiple Myeloma Treatment Market Snapshot, 2026 and 2033
- Market Opportunity Assessment, 2026 - 2033, US$ Bn
- Key Market Trends
- Future Market Projections
- Premium Market Insights
- Industry Developments and Key Market Events
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definition
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Key Trends
- Macro-economic Factors
- Global Sectoral Outlook
- Global GDP Growth Outlook
- COVID-19 Impact Analysis
- Forecast Factors - Relevance and Impact
- Value Added Insights
- Tool Adoption Analysis
- Regulatory Landscape
- Value Chain Analysis
- PESTLE Analysis
- Porter’s Five Force Analysis
- Price Analysis, 2025A
- Key Highlights
- Key Factors Impacting Deployment Costs
- Pricing Analysis, By Treatment Type
- Global Multiple Myeloma Treatment Market Outlook
- Key Highlights
- Market Volume (Units) Projections
- Market Size (US$ Bn) and Y-o-Y Growth
- Absolute $ Opportunity
- Market Size (US$ Bn) and Volume (Units) Analysis and Forecast
- Historical Market Size (US$ Bn) Analysis, 2020-2025
- Current Market Size (US$ Bn) Analysis and Forecast, 2026 - 2033
- Global Multiple Myeloma Treatment Market Outlook: Treatment Type
- Introduction / Key Findings
- Historical Market Size (US$ Bn) and Volume (Units) Analysis, By Treatment Type, 2020 - 2025
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Treatment Type, 2026 - 2033
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Stem Cell Transplant
- Radiation Therapy
- Market Attractiveness Analysis: Treatment Type
- Global Multiple Myeloma Treatment Market Outlook: Stage of Disease
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis, By Stage of Disease, 2020 - 2025
- Current Market Size (US$ Bn) Analysis and Forecast, By Stage of Disease, 2026 - 2033
- Stage I
- Stage II
- Stage III
- Market Attractiveness Analysis: Stage of Disease
- Global Multiple Myeloma Treatment Market Outlook: Drug Class
- Introduction / Key Findings
- Historical Market Size (US$ Bn) Analysis, By Drug Class, 2020 - 2025
- Current Market Size (US$ Bn) Analysis and Forecast, By Drug Class, 2026 - 2033
- Immunomodulatory Drugs
- Proteasome Inhibitors
- Monoclonal Antibodies
- CAR T-Cell Therapy
- Bispecific Antibodies
- Corticosteroids & Chemotherapy Agents
- Market Attractiveness Analysis: Drug Class
- Key Highlights
- Global Multiple Myeloma Treatment Market Outlook: Region
- Key Highlights
- Historical Market Size (US$ Bn) and Volume (Units) Analysis, By Region, 2020 - 2025
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Region, 2026 - 2033
- North America
- Europe
- East Asia
- South Asia and Oceania
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Multiple Myeloma Treatment Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Treatment Type
- By Stage of Disease
- By Drug Class
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- U.S.
- Canada
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Treatment Type, 2026 - 2033
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Stem Cell Transplant
- Radiation Therapy
- Current Market Size (US$ Bn) Analysis and Forecast, By Stage of Disease, 2026 - 2033
- Stage I
- Stage II
- Stage III
- Current Market Size (US$ Bn) Analysis and Forecast, By Drug Class, 2026-2033
- Immunomodulatory Drugs
- Proteasome Inhibitors
- Monoclonal Antibodies
- CAR T-Cell Therapy
- Bispecific Antibodies
- Corticosteroids & Chemotherapy Agents
- Market Attractiveness Analysis
- Europe Multiple Myeloma Treatment Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Treatment Type
- By Stage of Disease
- By Drug Class
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- Germany
- France
- U.K.
- Italy
- Spain
- Russia
- Türkiye
- Rest of Europe
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Treatment Type, 2026 - 2033
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Stem Cell Transplant
- Radiation Therapy
- Current Market Size (US$ Bn) Analysis and Forecast, By Stage of Disease, 2026 - 2033
- Stage I
- Stage II
- Stage III
- Current Market Size (US$ Bn) Analysis and Forecast, By Drug Class, 2026-2033
- Immunomodulatory Drugs
- Proteasome Inhibitors
- Monoclonal Antibodies
- CAR T-Cell Therapy
- Bispecific Antibodies
- Corticosteroids & Chemotherapy Agents
- Market Attractiveness Analysis
- East Asia Multiple Myeloma Treatment Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Treatment Type
- By Stage of Disease
- By Drug Class
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- China
- Japan
- South Korea
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Treatment Type, 2026 - 2033
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Stem Cell Transplant
- Radiation Therapy
- Current Market Size (US$ Bn) Analysis and Forecast, By Stage of Disease, 2026 - 2033
- Stage I
- Stage II
- Stage III
- Current Market Size (US$ Bn) Analysis and Forecast, By Drug Class, 2026-2033
- Immunomodulatory Drugs
- Proteasome Inhibitors
- Monoclonal Antibodies
- CAR T-Cell Therapy
- Bispecific Antibodies
- Corticosteroids & Chemotherapy Agents
- Market Attractiveness Analysis
- South Asia & Oceania Multiple Myeloma Treatment Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Treatment Type
- By Stage of Disease
- By Drug Class
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- India
- Southeast Asia
- ANZ
- Rest of South Asia & Oceania
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Treatment Type, 2026 - 2033
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Stem Cell Transplant
- Radiation Therapy
- Current Market Size (US$ Bn) Analysis and Forecast, By Stage of Disease, 2026 - 2033
- Stage I
- Stage II
- Stage III
- Current Market Size (US$ Bn) Analysis and Forecast, By Drug Class, 2026-2033
- Immunomodulatory Drugs
- Proteasome Inhibitors
- Monoclonal Antibodies
- CAR T-Cell Therapy
- Bispecific Antibodies
- Corticosteroids & Chemotherapy Agents
- Market Attractiveness Analysis
- Latin America Multiple Myeloma Treatment Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Treatment Type
- By Stage of Disease
- By Drug Class
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- Brazil
- Mexico
- Rest of Latin America
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Treatment Type, 2026 - 2033
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Stem Cell Transplant
- Radiation Therapy
- Current Market Size (US$ Bn) Analysis and Forecast, By Stage of Disease, 2026 - 2033
- Stage I
- Stage II
- Stage III
- Current Market Size (US$ Bn) Analysis and Forecast, By Drug Class, 2026-2033
- Immunomodulatory Drugs
- Proteasome Inhibitors
- Monoclonal Antibodies
- CAR T-Cell Therapy
- Bispecific Antibodies
- Corticosteroids & Chemotherapy Agents
- Market Attractiveness Analysis
- Middle East & Africa Multiple Myeloma Treatment Market Outlook
- Key Highlights
- Historical Market Size (US$ Bn) Analysis, By Market, 2020 - 2025
- By Country
- By Treatment Type
- By Stage of Disease
- By Drug Class
- Current Market Size (US$ Bn) Analysis and Forecast, By Country, 2026 - 2033
- GCC Countries
- Egypt
- South Africa
- Northern Africa
- Rest of Middle East & Africa
- Current Market Size (US$ Bn) and Volume (Units) Analysis and Forecast, By Treatment Type, 2026 - 2033
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Stem Cell Transplant
- Radiation Therapy
- Current Market Size (US$ Bn) Analysis and Forecast, By Stage of Disease, 2026 - 2033
- Stage I
- Stage II
- Stage III
- Current Market Size (US$ Bn) Analysis and Forecast, By Drug Class, 2026-2033
- Immunomodulatory Drugs
- Proteasome Inhibitors
- Monoclonal Antibodies
- CAR T-Cell Therapy
- Bispecific Antibodies
- Corticosteroids & Chemotherapy Agents
- Market Attractiveness Analysis
- Competition Landscape
- Market Share Analysis, 2025
- Market Structure
- Competition Intensity Mapping By Market
- Competition Dashboard
- Company Profiles (Details - Overview, Financials, Strategy, Recent Developments)
- Bristol-Myers Squibb
- Overview
- Segments and Deployments
- Key Financials
- Market Developments
- Market Strategy
- Takeda Pharmaceutical Company
- Johnson & Johnson
- Amgen
- AbbVie
- Celgene
- Pfizer
- Sanofi
- Gilead Sciences
- Regeneron Pharmaceuticals
- Roche
- Karyopharm Therapeutics
- BeiGene
- Legend Biotech
- Bristol-Myers Squibb
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations
- Pharmaceuticals
- Multiple Myeloma Treatment Market
Multiple Myeloma Treatment Market Size, Share, Trends, Growth, Regional Forecasts, 2026 - 2033
Multiple Myeloma Treatment Market by Treatment Type (Chemotherapy, Immunotherapy, Targeted Therapy, Stem Cell Transplant, Radiation Therapy), Stage of Disease (Stage I, Stage II, Stage III), Drug Class (Immunomodulatory Drugs, Proteasome Inhibitors, Monoclonal Antibodies, CAR T Cell Therapy, Bispecific Antibodies, Corticosteroids & Chemotherapy Agents) and Regional Analysis for 2026 - 2033
Key Industry Highlights
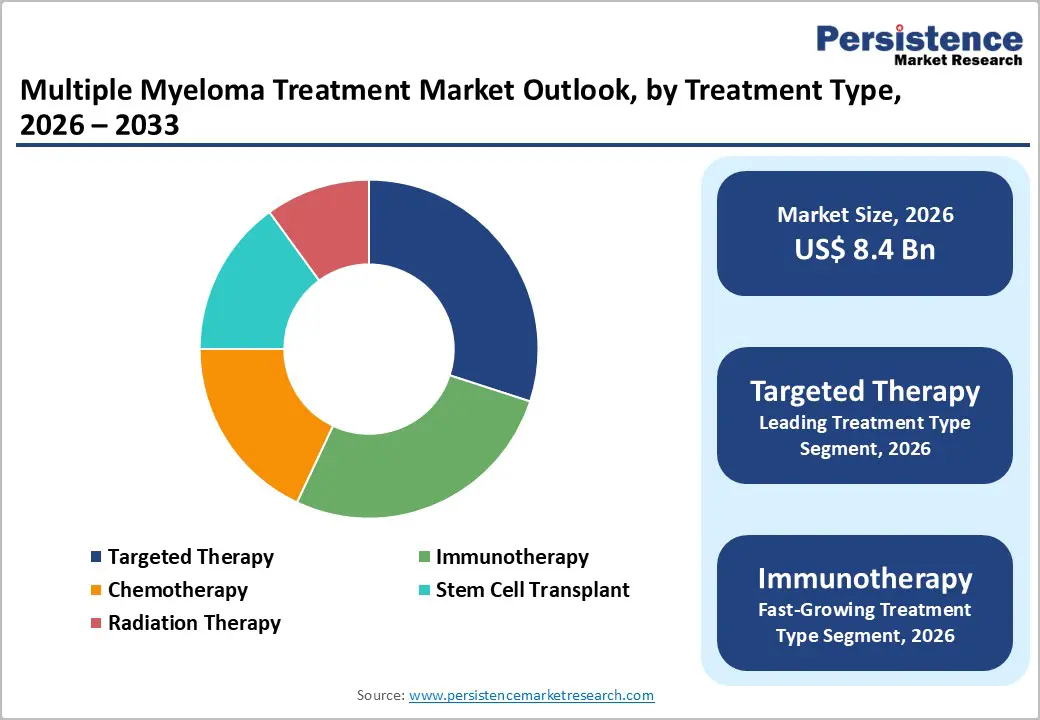
- Leading Treatment Type: Targeted therapy is projected to hold the largest revenue share of nearly 30% in 2026, due to their clinical efficacy and hospital adoption.
- Fastest-Growing Treatment Type: Immunotherapy is expected to grow the fastest between 2026 and 2033, driven by CAR-T therapies and the adoption of monoclonal antibodies.
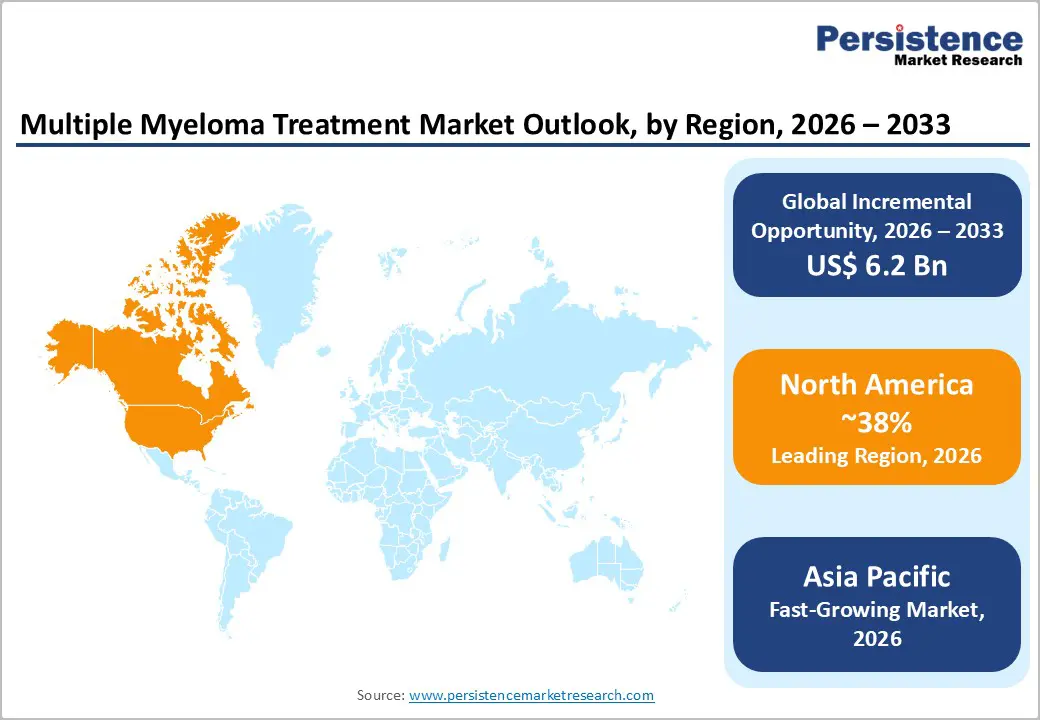
- Dominant Region: North America is expected to dominate with an estimated 38% market share in 2026, supported by advanced healthcare infrastructure.
- Fastest-Growing Regional Market: Asia Pacific is forecast to be the fastest-growing market through 2033, driven by rising private healthcare investment and digital health adoption.
- Competitive Environment: The market is moderately consolidated, led by multinational pharmaceutical companies with strong R&D, partnerships, and differentiated products.
| Key Insights | Details |
|---|---|
|
Multiple Myeloma Treatment Market Size (2026E) |
US$ 8.4 Bn |
|
Market Value Forecast (2033F) |
US$ 14.6 Bn |
|
Projected Growth (CAGR 2026 to 2033) |
8.2% |
|
Historical Market Growth (CAGR 2020 to 2025) |
6.5% |
DRO Analysis
Growing Number of Multiple Myeloma Cases in Aging Populations
High incidence of multiple myeloma in older age groups directly increases demand for diagnosis and treatment. In the United States, new cases are projected at 36,110 in 2025, with most diagnoses occurring in people aged 65 and above, while diagnoses in younger adults remain rare. The U.S. Centers for Disease Control and Prevention (CDC) identifies age as the most significant risk factor, noting that individuals under 45 years rarely develop this disease and that risk rises sharply with age. An older demographic typically has more comorbidities and a slower immune response, which increases complication rates and care needs.
Healthcare systems face greater pressure for specialized oncology care as the population ages. Treatment complexity and long-term management increase resource use, driving demand for novel therapies and expanded clinical services. With older adults representing a growing share of cancer cases, payers and providers prioritize investments in treatment pathways and supportive care. This demographic shift also influences funding for clinical research and therapy innovation, as regulators and healthcare planners adjust to manage a larger, older patient population that requires comprehensive, long-term care solutions.
Increasing Investment in New Drug Development
Growing government and public funding for research drives new drug development as a key driver of innovation. Public agencies, such as the FY 2025 National Cancer Institute Congressional Justification Fact Sheet, invest billions to support grants that explore novel therapeutic targets and clinical approaches. For example, the United States’ National Cancer Institute (NCI) had over $7.2 billion allocated to research and grants supporting cancer discovery in fiscal year 2025, enabling work on plasma cell cancers such as multiple myeloma. Public funding underpins early-stage research, translational studies, and clinical trials that pharmaceutical companies build on, reducing barriers to science that can lead to new molecular therapies or immunotherapies.
The need for more effective treatment options creates strong incentives for this investment. Traditional therapies often deliver limited benefit for relapsed patients, so research aims to overcome resistance and improve outcomes. Government-backed research projects, including those listed on National Institutes of Health (NIH) databases with FY-2025 funding for specific drug resistance studies, illustrate targeted efforts to understand disease biology and develop next-generation agents. Public financing supports collaborations between academia and industry, accelerates the availability of clinical trials, and reduces risk for private investors. This broad funding landscape expands the pipeline of novel therapies and stimulates sustained investment in drug assets that can transform clinical care pathways.
Limited Specialized Treatment Centers
Access to advanced multiple myeloma therapies remains uneven across regions due to a shortage of specialized treatment centers. These centers require high levels of expertise, advanced infrastructure, and multidisciplinary teams capable of administering complex therapies such as CAR-T cell treatments and stem cell transplants. Patients in rural or underdeveloped areas face significant travel and logistical challenges to reach such facilities, resulting in delayed treatment initiation and reduced adherence to prescribed regimens. The concentration of specialized centers in urban or high-income regions creates disparities in patient outcomes and limits overall market penetration for innovative therapies.
Establishing new specialized centers demands substantial capital investment, skilled personnel, and strict regulatory compliance. Training programs for oncologists, nurses, and laboratory staff are intensive, extending the timeline for operational readiness. Smaller hospitals and clinics lack the resources to support the technical requirements of high-cost therapies, including advanced laboratory diagnostics, cryopreservation, and post-treatment monitoring. These operational and financial constraints restrict expansion into underserved areas, slowing the adoption of emerging treatment protocols.
High Cost of Advanced Treatments
The pricing of multiple myeloma therapies remains a significant market restraint due to the complexity of drug development and production. Innovative treatments such as CAR-T cell therapies, monoclonal antibodies, and bispecific antibodies involve extensive research, clinical trials, and specialized manufacturing processes. These factors drive substantial capital investment, resulting in elevated treatment costs. Reimbursement policies vary across regions, creating affordability gaps for patients and limiting the adoption of treatments. Limited competition in certain therapy segments further maintains price levels. The financial burden affects healthcare providers and payers, influencing decisions on treatment protocols and patient coverage.
Logistical and operational requirements also contribute to cost pressures. Advanced therapies require specialized infrastructure, cold-chain storage, and trained personnel for administration, thereby increasing operational expenses for hospitals and clinics. Market entry of novel therapies remains slow due to stringent regulatory approvals and high investment thresholds. Patient out-of-pocket expenses influence access and adherence, particularly in regions with limited insurance coverage. Pricing challenges restrict widespread adoption and market penetration, impacting overall growth dynamics and shaping strategic planning for stakeholders across the multiple myeloma treatment landscape.
Growth of Targeted and Personalized Therapies
Targeted and personalized therapies represent a key opportunity because they can precisely attack cancer cells based on specific disease characteristics. These therapies focus on molecular features specific to each patient’s disease and improve treatment effectiveness while minimizing impact on healthy cells. For example, CD38-targeting monoclonal antibodies recently expanded treatment options, leading to longer progression-free survival in newly diagnosed patients compared with traditional regimens, with significantly improved outcomes reported in 2025 by a U.S. government cancer research site.
The rationale for prioritizing these therapies lies in the heterogeneity of disease biology. Standard chemotherapies act broadly, often harming normal cells and limiting dose intensity. Personalized approaches use genetic and protein biomarkers to identify optimal targets and match patients with therapies most likely to be effective. Ongoing development of bispecific antibodies and engineered cell therapies extends treatment options for refractory patients and addresses unmet needs in subgroups with high unmet medical need. Continued investment in these innovations is expected to shape future therapeutic paradigms.
Use of Digital Tools for Monitoring and Diagnosis
The increasing access to digital health records and tools is a key opportunity because it directly supports more frequent and accurate monitoring of patient status and facilitates timely clinical decisions. In the U.S., by 2024 about 75% of individuals managing a recent cancer diagnosis were offered and accessed their online medical records or patient portal via apps or web-based interfaces, highlighting widespread acceptance and use of digital platforms for health management. This access enables patients and clinicians to view test results, treatment updates, and symptom reports in real time, thereby improving monitoring, supporting early recognition of disease progression or complications, and reducing delays in care delivery.
Government policy, through the 21st Century Cures Act and related standards, has accelerated access to electronic health information, with many hospitals adopting patient engagement capabilities that enable secure messaging and the electronic sharing of clinical data. Digital tools also empower patients to track symptoms, report outcomes between visits, and engage more actively in their care, leading to richer datasets that clinicians can use to personalize treatment adjustments and strengthen patient–provider collaboration.
Category-wise Analysis
Treatment Type Insights
Targeted therapy is likely to claim nearly 30% of the multiple myeloma treatment market revenue share in 2026, due to its high clinical efficacy and precision in treating multiple myeloma. These therapies selectively inhibit molecular pathways essential for cancer proliferation, reducing damage to healthy cells and improving safety profiles. Oncology centers prioritize targeted therapy for first-line and relapsed patients, guided by clinical evidence and trial outcomes. Combination regimens with immunomodulatory drugs or proteasome inhibitors enhance effectiveness, while provider expertise and accessibility in developed regions support broad adoption.
Immunotherapy is expected to witness the fastest growth between 2026 and 2033, driven by advancements in monoclonal antibodies, CAR-T cell therapies, and bispecific antibodies. Adoption rises due to improved relapse-free survival and patient-specific targeting. Expansion of specialized clinical infrastructure, including infusion centers, supports delivery and monitoring. Real-world evidence demonstrates clinical benefits, increasing provider confidence. Regulatory agencies in North America and Europe enable faster approvals, while growing awareness among oncologists and patients and rising healthcare access in urban and emerging markets fuel market growth.
Drug Class Insights
Immunomodulatory drugs are poised to lead, with a forecasted over 35% market share in 2026, owing to broad clinical acceptance and integration into combination therapy regimens. These drugs enhance immune system activity, inhibit tumor proliferation, and are incorporated into multiple standard-of-care protocols. Their use spans first-line and relapsed patient populations, driving consistent adoption. Combination strategies with proteasome inhibitors or targeted therapies further improve treatment outcomes. Widespread familiarity among healthcare providers and inclusion in clinical guidelines reinforce their preference in oncology centers, sustaining revenue growth.
CAR-T cell therapy is anticipated to be the fastest-growing segment between 2026 and 2033, driven by innovation in personalized cell-based therapies and regulatory facilitation. Adoption is supported by strong clinical outcomes in relapsed or refractory multiple myeloma cases. The expansion of specialized oncology centers with trained personnel enables effective delivery of therapy. Collaborations between biotechnology companies and healthcare providers improve scalability and access. Rising awareness among clinicians and patients, along with increasing reimbursement coverage in developed markets, contributes to rapid market penetration and adoption of CAR-T therapies.
Regional Insights
North America Multiple Myeloma Treatment Market Trends and Insights
North America is expected to lead with an estimated 38% of the multiple myeloma treatment market share in 2026, supported by extensive clinical trial activity and advanced healthcare infrastructure in the United States and Canada. Widespread adoption of CAR-T cell treatments, bispecific antibodies, and targeted therapies drives high treatment penetration. Well-capitalized hospitals and oncology centers enable rapid deployment of complex regimens. Comprehensive insurance coverage improves patient access to costly therapies. Continuous investment in research and development accelerates the commercialization of novel drugs. Professional training and evidence-based clinical protocols reinforce provider confidence, increasing utilization across diverse patient populations.
Significant investment in personalized therapy development sustains leadership in multiple myeloma care in the United States and Canada. Advanced diagnostic capabilities and specialized infusion facilities enable the implementation of precision medicine strategies. Collaborations between biotechnology firms, hospitals, and research institutes accelerate therapy availability and operational efficiency. Early integration of regulatory-approved innovations into standard care promotes adoption of novel protocols. Aging populations are driving demand for early intervention and combination regimens. Data-driven decision-making and real-world outcome monitoring strengthen clinical confidence, ensuring consistent market dominance.
Europe Multiple Myeloma Treatment Market Trends and Insights
Europe is projected to hold a prominent position in the market for multiple myeloma treatment, due to advanced healthcare infrastructure and clinical research networks in Germany and France. High adoption of targeted therapies, immunomodulatory drugs, and CAR-T cell treatments drives patient access. Well-structured reimbursement frameworks support uptake of high-cost therapies. Expansion of specialized oncology centers and infusion units enables implementation of complex regimens. Robust clinical trials generate real-world evidence, strengthening provider confidence. Continuous professional training optimizes patient management and reinforces therapy utilization.
Innovation and regulatory support enhance adoption in the United Kingdom and Italy. Collaborations between pharmaceutical companies and hospitals facilitate commercialization of advanced therapies. Integration of combination regimens and precision medicine improves outcomes. Aging populations and rising incidence increase demand for early diagnosis and comprehensive treatment strategies. Investments in digital health tools support monitoring and adherence. Evidence-based guidelines, centralized healthcare systems, and early access programs accelerate adoption of novel therapies across multiple treatment segments.
Asia Pacific Multiple Myeloma Treatment Market Trends and Insights
Asia Pacific is forecasted to be the fastest-growing market for multiple myeloma treatment between 2026 and 2033, stimulated by rising investment in healthcare infrastructure and oncology centers in China and India. The expansion of specialized infusion units and diagnostic facilities enables the implementation of advanced therapies. Growing awareness of early detection and disease management drives adoption of targeted therapies and immunotherapies. Government initiatives to improve cancer care and reimbursement schemes support patient access. Urbanization and rising disposable income increase treatment affordability. Biopharmaceutical partnerships accelerate the introduction of novel drugs. Clinical training programs enhance provider expertise, strengthening therapy utilization across large patient populations.
Rapid adoption of innovative therapies is observed in Japan and South Korea due to advanced healthcare systems and strong regulatory support. Oncology centers integrate CAR-T cell therapies, bispecific antibodies, and immunomodulatory regimens into standard protocols. The expansion of clinical trial networks and real-world outcome monitoring facilitate evidence-based adoption. Investments in biotechnology firms and hospital collaborations enhance therapy delivery and scalability. Aging populations drive demand for complex treatment regimens. Increasing digital health initiatives support early diagnosis and patient monitoring, strengthening infrastructure and accelerating market penetration in both countries.
Competitive Landscape
The global multiple myeloma treatment market is moderately consolidated, with leading players capturing a significant share of global revenue. Bristol-Myers Squibb, Takeda Pharmaceutical Company, Johnson & Johnson, and Amgen maintain strong market influence through diversified portfolios including immunomodulatory drugs, CAR-T therapies, and targeted agents. Market dominance is reinforced by high entry barriers such as regulatory approvals, extensive research and development, and complex manufacturing requirements for biologics and cell-based therapies, limiting competition from smaller companies.
Competitive positioning relies on product differentiation, clinical efficacy, and geographic reach. Strategic partnerships and collaborations enable faster commercialization and global distribution. Mid-sized biotech firms contribute through specialized innovations and niche therapies, often collaborating with larger players to expand market access. Adoption of combination regimens, personalized treatment strategies, and real-world clinical evidence further strengthens the leadership of major companies, maintaining sustained influence and shaping treatment protocols in multiple myeloma care.
Key Industry Developments
- In March 2026, researchers at Emory University reported that multiple myeloma treatment paradigms progressed toward quadruple frontline regimens and minimal residual disease testing, improving deep response rates and informing relapse strategies including BCMA-targeted therapies and genomic profiling.
- In February 2026, researchers at Technical University of Munich showed that an established proteasome inhibitor drug can reactivate CAR-T immunotherapy in multiple myeloma patients whose treatment had stopped working, restoring target antigen expression and improving therapy response.
- In January 2026, researchers at Memorial Sloan Kettering Cancer Center reported that expanded use of novel drug combinations and immunotherapies, including recent U.S. Food and Drug Administration (FDA)-approved monoclonal antibody regimens, has significantly improved prognosis and extended survival for people living with multiple myeloma.
Companies Covered in Multiple Myeloma Treatment Market
- Bristol-Myers Squibb
- Takeda Pharmaceutical Company
- Johnson & Johnson
- Amgen
- AbbVie
- Celgene
- Pfizer
- Sanofi
- Gilead Sciences
- Regeneron Pharmaceuticals
- Roche
- Karyopharm Therapeutics
- BeiGene
- Legend Biotech
Frequently Asked Questions
The global multiple myeloma treatment market is projected to reach US$ 8.4 billion in 2026.
Rising prevalence among aging populations and adoption of advanced therapies are driving the market.
The market is poised to witness a CAGR of 8.2% from 2026 to 2033.
Expansion of personalized therapies, immunotherapies, and emerging market access presents key opportunities in the market.
Some of the key market players include Bristol-Myers Squibb, Takeda Pharmaceutical Company, Johnson & Johnson, and Amgen.













