
- Biotechnology
- Gene Switch Market
Gene Switch Market Size, Share, and Growth Forecast, 2026 - 2033
Gene Switch Market by Product Type (Inducible Gene Switches, Repressible Gene Switches), Technology (CRISPR-based Systems, Zinc Finger Proteins (ZFP), Others), Application (Gene Therapy, Synthetic Biology, Others), and Regional Analysis for 2026 - 2033
Gene Switch Market Size and Trends Analysis
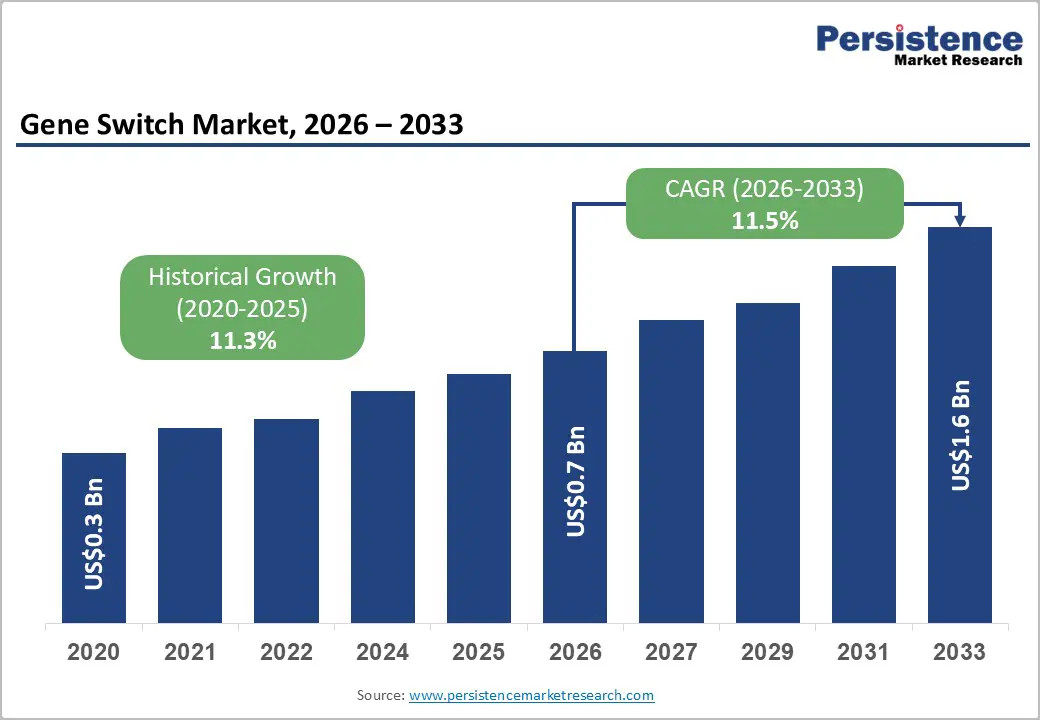
The global gene switch market size is likely to be valued at US$0.7 billion in 2026 and is expected to reach US$1.6 billion by 2033, growing at a CAGR of 11.5% during the forecast period from 2026 to 2033, driven by recent, verifiable developments in the broader gene and cell therapy ecosystem, particularly highlighted by global health and research organizations.
According to the World Health Organization 2024 data, the field of cell and gene therapies witnessed significant acceleration, with over 1,900 ongoing clinical trials globally, primarily focused on oncology (over 50%) and rare genetic diseases, demonstrating the growing demand for controlled gene expression systems such as gene switches in advanced therapeutics.
According to insights from the National Institutes of Health (2025), continued investment in cancer and genetic disease research has significantly improved treatment innovation, supporting the development of targeted and gene-based therapies that rely on precise regulatory systems such as gene switches. Continuous advancements in gene-editing platforms, especially CRISPR-based regulatory systems, along with the integration of programmable biology and automation, are significantly improving the efficiency and scalability of these technologies.
Key Industry Highlights:
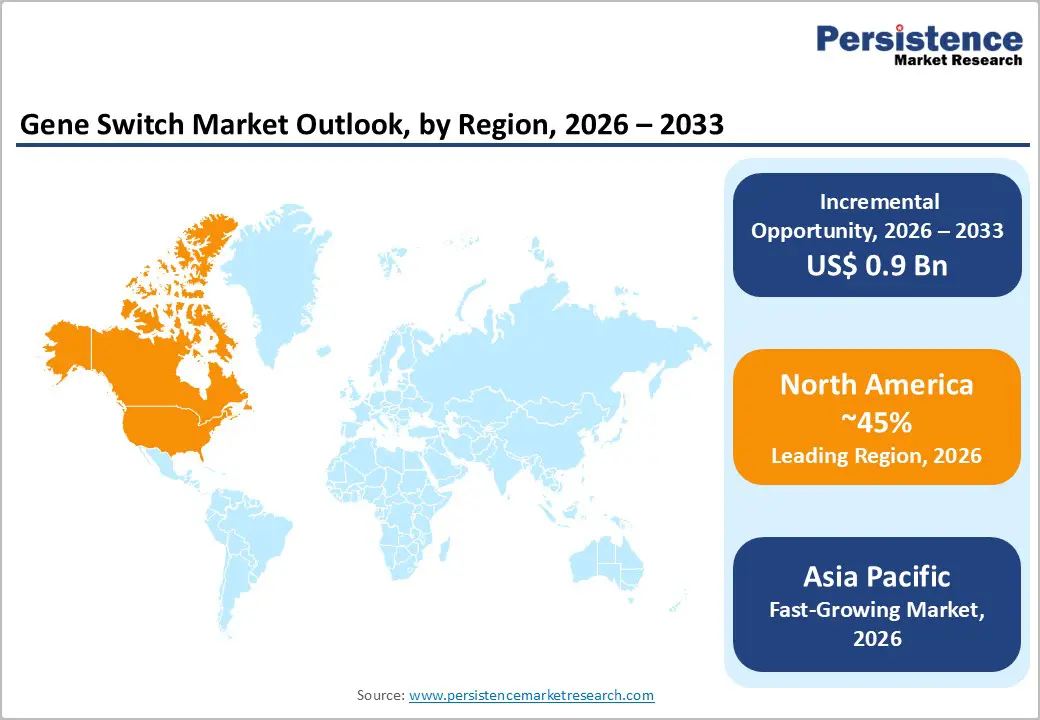
- Leading Region: North America is anticipated to be the leading region, accounting for a market share of 45% in 2026, driven by strong biotechnology innovation, advanced regulatory frameworks, and high adoption of gene and cell therapies.
- Fastest-growing Region: Asia Pacific is likely to be the fastest-growing region, supported by rapid biotech expansion and strong government support.
- Leading Product Type: Inducible gene switches are projected to represent the leading product type in 2026, accounting for 60% of the revenue share, driven by their flexible activation control in therapeutic applications.
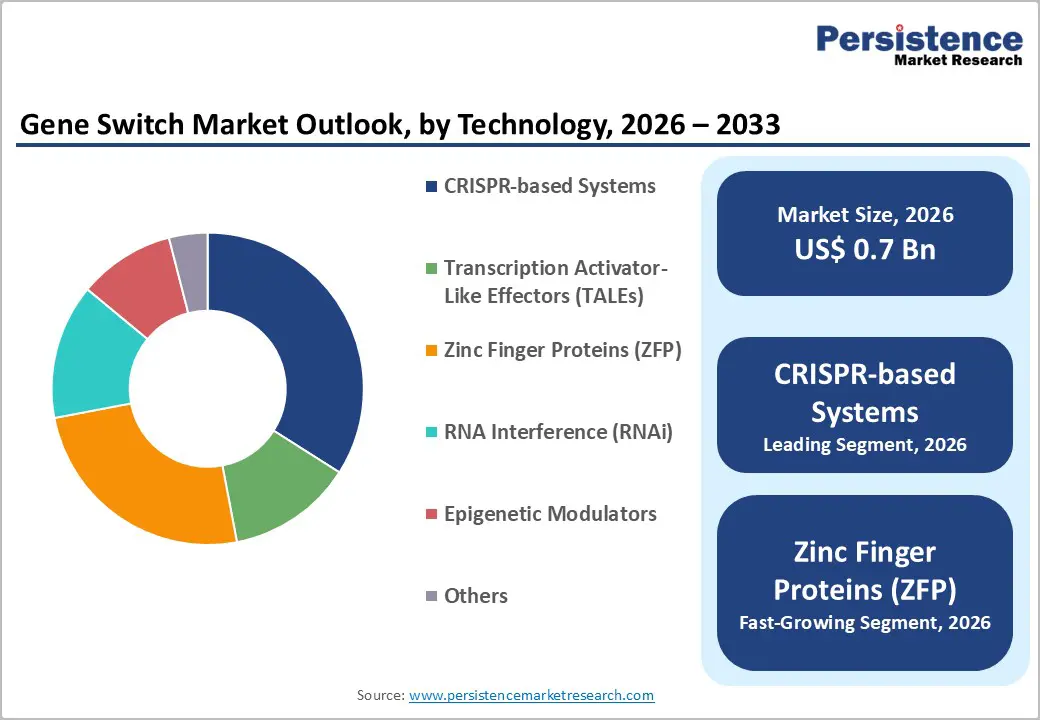
- Leading Technology: CRISPR-based systems are anticipated to be the leading technology, accounting for over 50% of the revenue share in 2026, supported by high programmability and widespread adoption in gene regulation.
| Key Insights | Details |
|---|---|
| Gene Switch Market Size (2026E) | US$0.7 Bn |
| Market Value Forecast (2033F) | US$1.6 Bn |
| Projected Growth (CAGR 2026 to 2033) | 11.5% |
| Historical Market Growth (CAGR 2020 to 2025) | 11.3% |
DRO Analysis
Driver - Advancements in Gene and Cell Therapy Safety Requirements
The growing complexity of gene and cell therapies has significantly increased the need for precise safety control mechanisms, positioning gene switches as critical components in modern therapeutic design. Advanced treatments such as CAR-T cell therapies require controlled activation and deactivation to prevent adverse effects such as cytokine release syndrome and off-target toxicity. Gene switches enable temporal and spatial regulation of therapeutic genes, allowing clinicians to modulate treatment intensity based on patient response.
Regulatory agencies are increasingly emphasizing safety and controllability in advanced therapy approvals, driving the integration of gene switch technologies. The ability to incorporate kill switches or reversible activation systems into engineered cells aligns with stringent clinical safety requirements. Advancements in inducible systems using well-characterized molecules improve predictability and reduce risks during clinical trials. As personalized medicine expands, the need for adaptive therapeutic control continues to grow.
Technological Convergence with CRISPR and Synthetic Biology Tools
The integration of gene switch systems with advanced genome-editing platforms has transformed the landscape of gene regulation, enabling highly programmable and efficient biological systems. CRISPR-based tools allow precise targeting of genetic sequences, while synthetic biology frameworks enable the design of complex gene circuits capable of executing logical functions. This convergence enhances the scalability and versatility of gene switches, supporting applications in therapeutics, industrial biotechnology, and research.
Continuous innovation in synthetic biology is enabling the development of multi-layered gene circuits that can respond to environmental signals and cellular conditions in real time. These advancements facilitate the creation of sophisticated therapeutic systems capable of dynamic regulation, improving treatment precision. The decreasing cost of genome engineering tools and the availability of modular biological components support widespread adoption.
Restraint - Technological and Implementation Complexity
The development and deployment of gene switch systems involve significant technical challenges, including complex design requirements, integration with delivery systems, and maintaining stability within biological environments. Achieving precise control over gene expression requires careful optimization of multiple components, such as promoters, regulators, and inducers. Variability in cellular responses and differences in biological systems complicate implementation. These challenges increase development timelines and costs, limiting accessibility for smaller research organizations and delaying commercialization in clinical settings where consistency and reproducibility are critical.
Scaling gene switch technologies from laboratory research to clinical and industrial applications presents obstacles. Manufacturing consistency, regulatory compliance, and quality control require advanced infrastructure and expertise. Integration with delivery methods such as viral vectors or nanoparticles adds another layer of complexity. The need for interdisciplinary collaboration across molecular biology, engineering, and clinical sciences can slow progress. Addressing these challenges requires continued innovation, standardized platforms, and improved design tools to simplify development processes and enable broader adoption across therapeutic and industrial applications.
Limited Long-Term Clinical Data
The relatively recent emergence of gene switch technologies in clinical applications means that long-term safety and efficacy data remain limited. While early-stage trials demonstrate promising results, there is still uncertainty regarding sustained performance, potential immune responses, and long-term genetic stability. This lack of comprehensive longitudinal data can create hesitation among healthcare providers and regulatory bodies, slowing the adoption of gene switch-enabled therapies. Ensuring consistent outcomes over extended periods is particularly important for chronic conditions and lifelong treatments where gene regulation must remain stable and predictable.
Regulatory approval processes often require extensive clinical evidence to validate new technologies, and the absence of long-term data can lead to prolonged evaluation timelines. Potential risks such as unintended gene interactions or delayed adverse effects must be thoroughly assessed. Pharmaceutical companies may face challenges in securing investment and partnerships without robust clinical validation. To overcome these barriers, ongoing clinical trials and post-market surveillance are essential.
Opportunity - Unmet Needs in Safety-Enhanced Cell Therapies
The increasing adoption of cell-based therapies has highlighted critical gaps in safety and controllability, creating strong demand for gene switch technologies. Many current treatments lack precise mechanisms to regulate therapeutic activity once administered, leading to potential risks such as overactivation or unintended side effects. Gene switches address these challenges by providing real-time control over engineered cells, enabling clinicians to adjust or terminate therapy if necessary. This capability is particularly important in oncology and rare disease treatments, where patient-specific responses require adaptive and controlled therapeutic interventions.
As the pipeline of advanced therapies continues to expand, the need for integrated safety solutions becomes more urgent. Gene switches offer a strategic advantage by enhancing the reliability and flexibility of cell therapies, making them more acceptable to regulators and healthcare providers. The growing focus on personalized medicine amplifies this opportunity, as treatments must be tailored to individual patient profiles. By addressing critical safety concerns, gene switch technologies are positioned to play a central role in the next generation of therapeutic innovations and clinical advancements.
Technological Convergence with Optogenetics and Remote-Control Systems
Emerging technologies such as optogenetics and remote-controlled gene regulation systems are opening new avenues for innovation in the gene switch market. Optogenetic systems use light to precisely control gene expression, offering high spatial and temporal resolution without the need for chemical inducers. This approach enables non-invasive and reversible control, making it highly attractive for research and therapeutic applications. Integration with wearable or implantable devices enhances the potential for real-time monitoring and control of biological processes, improving treatment precision and patient outcomes.
The development of remote-controlled gene switches, including those activated by electromagnetic signals or ultrasound, represents a significant advancement in non-invasive therapy management. These technologies allow clinicians to modulate gene activity externally, reducing the need for repeated interventions. As research progresses, the combination of gene switches with advanced control systems is expected to enable highly personalized and responsive treatments.
Category-wise Analysis
Product Type Insights
Inducible gene switches are expected to lead, accounting for approximately 60% of revenue in 2026, driven by their high versatility in activating gene expression in response to external stimuli such as small molecules or environmental signals. Their leading position is supported by their widespread integration into gene and cell therapy pipelines, where precise control over therapeutic gene activation is essential for safety and efficacy. For example, their use in CAR-T cell therapies, where inducible switches help regulate immune cell activity and reduce adverse effects.
Repressible gene switches are likely to represent the fastest-growing segment, supported by demand increases for systems capable of turning gene expression “off” to enhance therapeutic safety. These switches play a crucial role in preventing overexpression and managing toxicity in engineered cells, particularly in long-term treatments. Growth is driven by the need for reversible and tightly controlled therapeutic mechanisms, especially in complex disease conditions. For example, their application as safety or “kill switches” in engineered immune cells, where gene activity can be suppressed to prevent harmful side effects.
Technology Insights
CRISPR-based systems are projected to lead the market, capturing around 50% of the revenue share in 2026, supported by their high precision, programmability, and ease of use in gene regulation and editing. These systems enable targeted control of gene expression through guide RNA-directed mechanisms and are widely adopted in both research and clinical applications. For example, the use of CRISPR-based switches in experimental gene therapies targeting genetic disorders, where precise activation or repression of specific genes is required.
Zinc finger proteins (ZFPs) are likely to be the fastest-growing technology, driven by their compact structure and suitability for applications requiring smaller genetic payloads. Despite competition from CRISPR systems, ZFPs offer advantages in specific therapeutic contexts, including targeted gene regulation with reduced delivery constraints. Growth is supported by advancements in protein engineering and increasing use in complex synthetic circuits. For example, their application in gene regulation therapies, where the delivery vector size is a limitation, makes ZFPs a practical alternative.
Regional Insights
North America Gene Switch Market Trends
North America is anticipated to be the leading region, accounting for a market share of 45% in 2026, driven by a strong biotechnology ecosystem, advanced research infrastructure, and high adoption of gene and cell therapies. The region benefits from significant funding for genomic research, increasing clinical trial activity, and rapid integration of advanced tools such as CRISPR-based regulatory systems. There is a growing emphasis on developing safer and more controllable therapeutic platforms, particularly for oncology and rare genetic disorders.
A key trend in the region is the increasing commercialization of programmable gene control platforms integrated into clinical-stage therapies. For example, Intellia Therapeutics is actively advancing CRISPR-based gene regulation approaches, demonstrating the potential of precise gene modulation in treating complex diseases. The region is also witnessing rising investments in synthetic biology and engineered cell therapies, enabling scalable production and improved treatment outcomes.
Europe Gene Switch Market Trends
Europe is likely to be a significant market, due to strong regulatory oversight, increasing investment in precision medicine, and a focus on safe and ethical deployment of advanced biotechnologies. The region places significant emphasis on compliance, clinical validation, and long-term safety, which influences the adoption of gene switch systems in therapeutic applications. Government-backed initiatives and funding programs are supporting research in synthetic biology and gene regulation, particularly in countries with established biotechnology sectors.
Europe is seeing the rise of collaborations between biotechnology firms and academic institutions to accelerate innovation and commercialization. For example, Sangamo Therapeutics has engaged in partnerships within the region to advance gene regulation technologies based on engineered DNA-binding proteins. These collaborations support the development of safer and more targeted therapies while aligning with stringent regulatory requirements.
Asia Pacific Gene Switch Market Trends
The Asia Pacific region is likely to be the fastest-growing region, driven by expanding biotechnology capabilities, increasing healthcare investments, and strong government support for innovation. Countries such as China and India are investing heavily in genomic research, infrastructure development, and clinical trial expansion. The region benefits from large patient populations and cost-effective manufacturing capabilities, which support the rapid adoption of gene-based technologies.
Asia Pacific is seeing the rise of domestic biotechnology companies focusing on gene editing and synthetic biology solutions tailored to regional needs. For example, CRISPR Therapeutics has expanded its footprint through collaborations and research initiatives that include engagement with Asia-Pacific markets. The region is also witnessing increased foreign direct investment and partnerships between multinational and local firms, enabling technology transfer and accelerating innovation.
Competitive Landscape
The global gene switch market exhibits a moderately fragmented structure, driven by the presence of multiple biotechnology innovators, emerging startups, and established pharmaceutical companies actively advancing gene regulation technologies. The competitive environment is shaped by rapid advancements in gene editing platforms, increasing clinical-stage pipelines, and strong intellectual property portfolios.
With key leaders including CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics, and Sangamo Therapeutics, the landscape reflects a mix of established expertise and emerging technological capabilities, supported by strong R&D investments and expanding clinical applications. These players compete through continuous innovation, strategic collaborations, and the development of proprietary gene switch platforms aimed at improving safety, specificity, and therapeutic efficiency.
Key Industry Developments:
- In November 2025, researchers at Weill Cornell Medicine, led by Dr. Samie Jaffrey, with Qian Hou as first author, developed a novel gene-switch technology called Cyclone, which enables safe, reversible control of gene activity using the antiviral drug acyclovir. The system leverages engineered “poison exons” to suppress gene expression until activated, allowing precise on/off regulation without altering RNA or causing toxicity.
- In September 2025, researchers led by Dr. Dubravka Vucicevic at the Max Delbrück Center, working in the lab of Professor Uwe Ohler, developed an advanced method called TESLA-seq to uncover previously hidden gene regulatory switches in human DNA. The technique combines CRISPR-based gene activation with targeted single-cell RNA sequencing, enabling faster and more accurate identification of transcriptional enhancers that control when and where genes are expressed.
Companies Covered in Gene Switch Market
- CRISPR Therapeutics
- Editas Medicine
- Sangamo Therapeutics
- Intellia Therapeutics
- Sana Biotechnology
Frequently Asked Questions
The global gene switch market is projected to reach US$0.7 billion in 2026.
The gene switch market is driven by the growing demand for precise, controllable, and safer gene and cell therapies enabled by advancements in gene regulation technologies.
The gene switch market is expected to grow at a CAGR of 11.5% from 2026 to 2033.
Key market opportunities lie in expanding applications of gene switches in precision medicine, advanced cell therapies, and integration with emerging technologies such as CRISPR and optogenetics.
CRISPR Therapeutics, Editas Medicine, Sangamo Therapeutics, and Intellia Therapeutics are the leading players.







