
- Pharmaceuticals
- Drug-Resistant Tuberculosis Market
Drug-Resistant Tuberculosis Market Size, Share, and Growth Forecast, 2026 - 2033
Drug-Resistant Tuberculosis Market by Drug Class (Amoxicillin/Clavulanate, Bedaquiline, Carbapenems with Clavulanic Acid, Clofazimine, Others), Disease Type (Active TB, Latent TB), Treatment Type, Distribution Channel, and Regional Analysis for 2026 - 2033
Drug-Resistant Tuberculosis Market Size and Trends Analysis
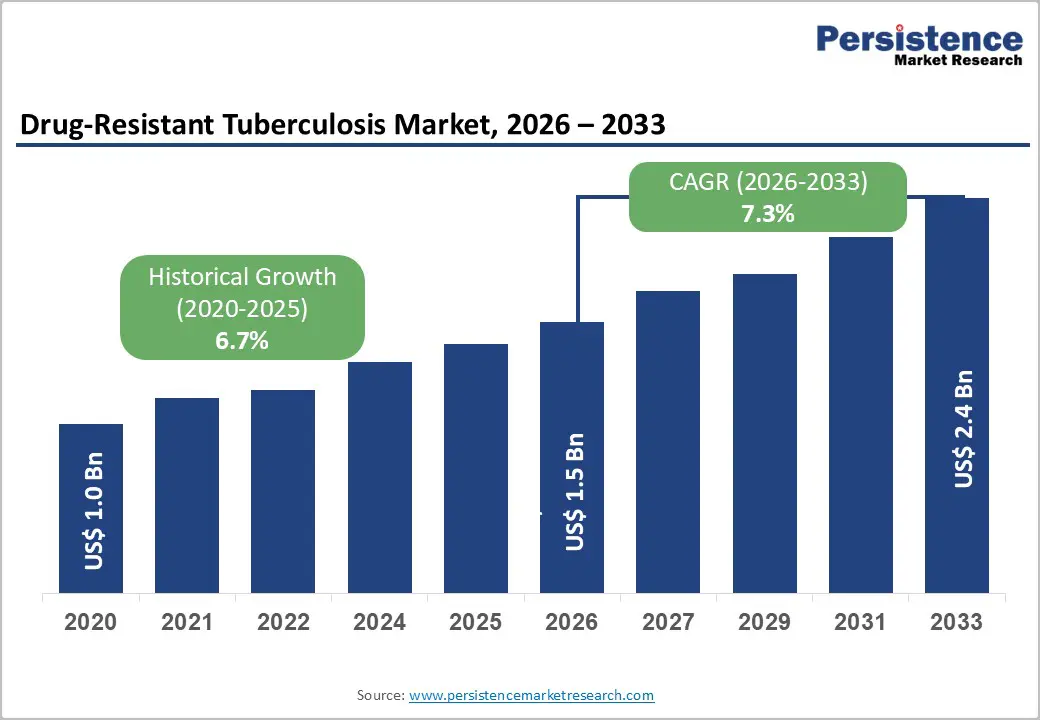
The global drug-resistant tuberculosis market size is likely to be valued at US$1.5 billion in 2026, and is expected to reach US$2.4 billion by 2033, growing at a CAGR of 7.3% during the forecast period from 2026 to 2033, driven by the rising prevalence of multidrug-resistant and extensively drug-resistant TB cases, increasing government initiatives for TB control, and the introduction of newer second-line therapies.
The increasing need for effective treatment options against resistant strains remains a major driver of the drug-resistant tuberculosis market growth.
Key Industry Highlights:
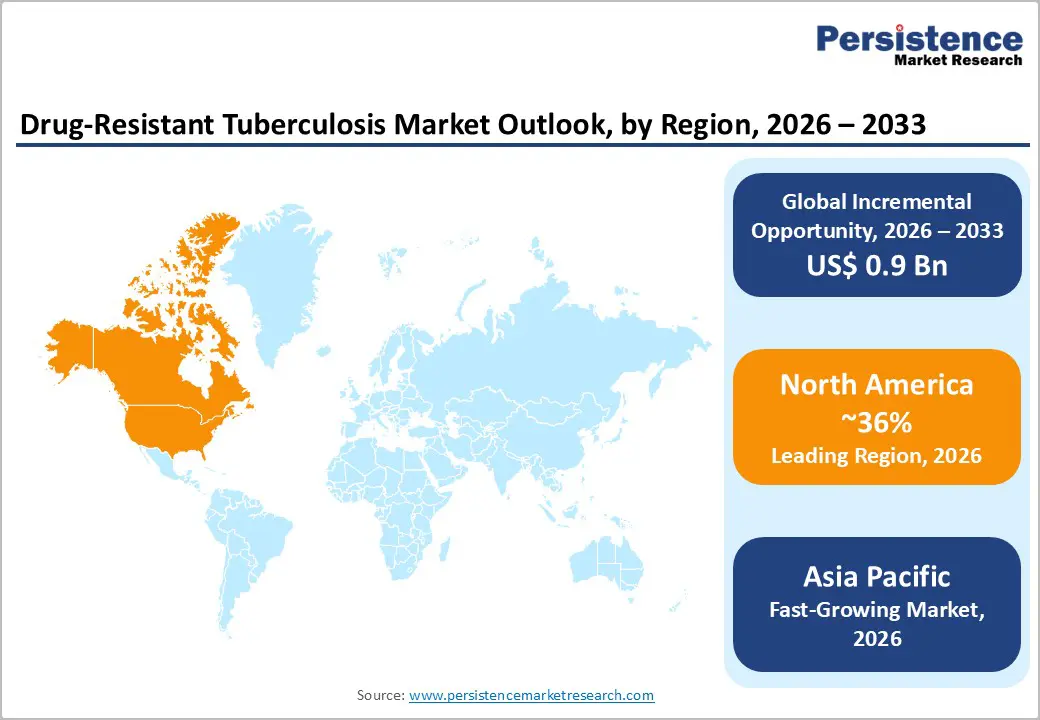
- Leading Region: North America, anticipated to account for a 36% market share in 2026, driven by early detection, precision treatment, and advanced diagnostics.
- Fastest-growing Region: Asia Pacific, fueled by early detection, advanced diagnostics, and expanded access to second-line therapies.
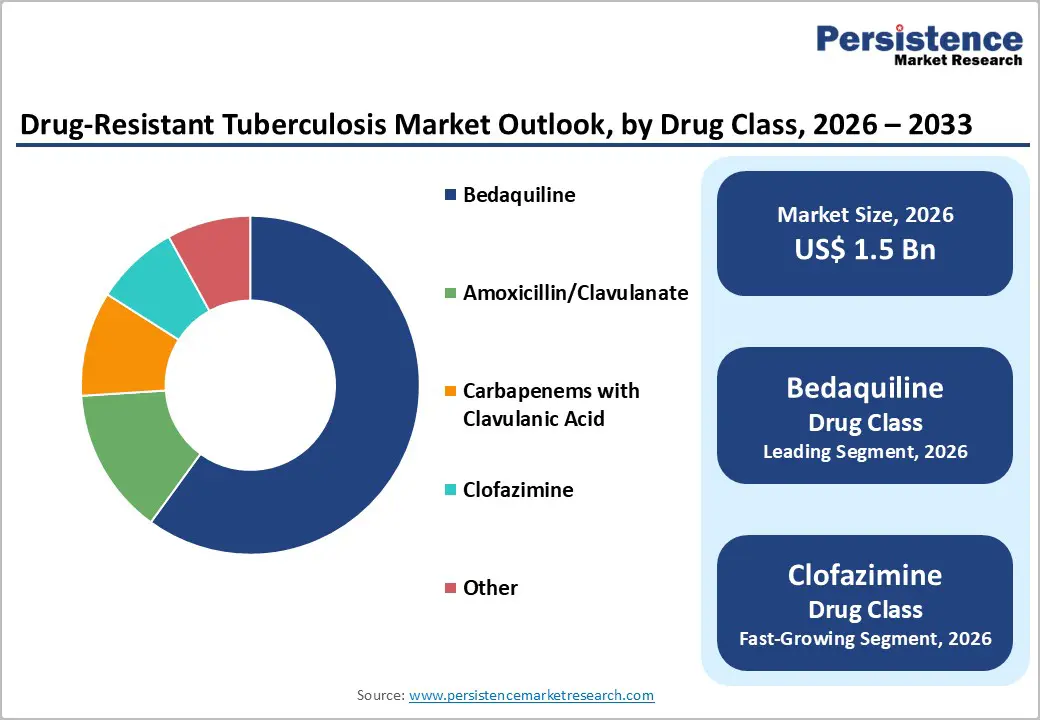
- Dominant Drug Class: Bedaquiline is expected to hold 60% market share in 2026, leading MDR-TB treatment with potent, oral, WHO-recommended regimens that improve adherence and outcomes.
- Leading Disease Type: Active TB is expected to dominate with 72% market share in 2026, driven by high treatment urgency and greater drug demand compared to latent TB.
| Key Insights | Details |
|---|---|
| Drug-Resistant Tuberculosis Market Size (2026E) | US$1.5 Bn |
| Market Value Forecast (2033F) | US$2.4 Bn |
| Projected Growth CAGR (2026 - 2033) | 7.3% |
| Historical Market Growth (2020 - 2025) | 6.7% |
DRO Analysis
Driver - Advancements in Drug Development
Recent progress in treatments for drug resistant tuberculosis reflects concerted global research and evidence based updates that aim to make therapy more effective, shorter, and better tolerated by patients. The World Health Organization (WHO) now recommends novel all oral regimens such as a six month combination that includes newer and repurposed medicines, moving away from lengthy injectable based therapy that was standard for MDR/RR TB (multidrug resistant or rifampicin resistant TB). This shift is based on results from large clinical trials like BEAT TB and endTB, which demonstrated strong evidence for the safety and efficacy of these regimens.
The global TB clinical development pipeline is expanding. As of 2025, there are nearly 29 drugs under investigation in various stages of clinical trials, including 18 new chemical entities, showing an acceleration in experimental therapies compared with a decade ago. Many of these are entirely new compounds designed specifically to overcome resistance, while others are being repurposed or combined in novel regimens to enhance their effectiveness and reduce treatment durations. These efforts are part of coordinated research platforms and investments aimed at diversifying and improving therapeutic options for drug resistant TB. Government and public health agencies such as the WHO and the CDC continue to support evidence driven strategies to integrate these advanced regimens into national treatment programs, emphasizing both efficacy and access so that more people can benefit from improved outcomes.
Rising Incidence of Drug Resistant TB
The burden of drug resistant tuberculosis (DRTB) continues to be a critical public health issue despite overall declines in some regions. According to the World Health Organization (WHO), an estimated 390,000 people developed multidrug resistant or rifampicin resistant TB (MDR/RRTB) in 2024, showing that resistant forms of TB remain a significant component of the global TB burden. Although the proportion of new TB cases with resistance has fallen over the past decade, resistant TB still accounts for a considerable share of the disease burden in high impact countries. In 2024, India alone accounted for around one third of these MDR/RR TB incident cases worldwide, highlighting the persistent challenge in high burden settings.
Regional patterns reveal variability, with increases in MDR/RR TB in areas such as the WHO Americas and South East Asia regions, even as other regions show declines. This indicates that trends may diverge locally due to factors such as healthcare access, diagnostics, treatment adherence, and social determinants of health.Drug resistant TB emerges primarily when TB bacteria evolve resistance to first line drugs, often through incomplete or inappropriate treatment, making it harder to cure and more costly to treat.
Restraint - High Cost of Treatment and Novel Drugs
Treatment of drug resistant tuberculosis (DR TB) poses a significant economic challenge for patients and health systems alike because the available regimens often involve newer and more expensive medications than those used for drug sensitive TB. Drugs such as bedaquiline, delamanid, and pretomanid represent important advances in therapy, but their higher production and procurement costs make them less affordable in many low and middle income countries where the DR TB burden is highest. Procurement expenses are further amplified by the need for combination therapies and longer durations of treatment, which drive up overall costs for national programs and insurers.
Government and donor programs, including the Global Drug Facility, work to negotiate reduced prices and supply support, but gaps remain in financing and access. Patients in settings without subsidized care often face out of pocket expenses for diagnostics, follow up, and supportive care, contributing to barriers to completing therapy. Moreover, the economic impact extends beyond direct medical costs; individuals with DR TB may experience loss of income due to prolonged illness, transportation costs, and caregiving responsibilities, exacerbating financial strain on households.
Complex, Long Duration Treatment Regimens
Treatment of drug resistant tuberculosis (DR TB) remains challenging due to the prolonged and complex nature of available regimens. Patients are often required to take multiple medications daily for many months, sometimes exceeding a year, which increases the risk of missed doses and incomplete therapy. The combination of several drugs is necessary to combat resistance, but it also raises the likelihood of side effects, including gastrointestinal disturbances, liver toxicity, and hearing loss, which can discourage adherence.
The lengthy duration of therapy places considerable strain on healthcare systems, requiring frequent monitoring, follow-up visits, and laboratory testing to ensure efficacy and manage adverse effects. Interruptions in treatment can lead to further drug resistance, complicating disease management and public health efforts. Health authorities, including the World Health Organization, emphasize the adoption of shorter, all-oral regimens where possible, which have shown improved patient adherence and treatment outcomes.
Opportunity- Expansion of Rapid and Point of Care Diagnostics
Rapid and point of care diagnostic testing has become a cornerstone in combating drug resistant tuberculosis (DR TB) because it allows healthcare providers to identify TB and its resistance patterns much earlier than traditional laboratory methods. Historically, diagnosis relied on sputum smear microscopy and culture, which can take weeks and often miss resistant strains, delaying appropriate treatment. In recent years, WHO recommended rapid molecular tests, such as nucleic acid amplification tests (NAATs), have transformed this landscape by providing highly specific and sensitive results within hours rather than days or weeks, enabling patients to begin proper therapy sooner. These tests can detect resistance to key anti TB drugs such as rifampicin and isoniazid at the time of initial diagnosis, which improves clinical decision making and reduces the period during which resistant TB can spread undetected.
In 2024, more than half of newly diagnosed TB patients were initially tested with such rapid tests, reflecting progress toward this goal, though substantial gaps remain in many countries and at peripheral health facilities. A key development in 2026 has been the WHO’s updated guidance endorsing near point of care tests and simpler diagnostic tools that can be used closer to where patients first seek care. These newer, portable tests are designed to bring accurate diagnosis into lower resource settings, shorten turnaround times, and reduce dependency on centralized laboratories, helping to catch DR TB cases sooner and initiate the right treatments faster.
Development of Novel and Shorter Treatment Regimens
Recent global guidance has shifted the treatment landscape for drug resistant tuberculosis (DR TB) by prioritizing shorter and more effective regimens that can improve outcomes and reduce the burden on patients and health systems. Historically, DR TB therapy often lasted 18-20 months or longer with multiple drugs and significant side effects. New recommendations by the World Health Organization (WHO) now include all oral regimens lasting about six months for eligible patients with multidrug resistant or rifampicin resistant TB (MDR/RR TB) and for some with additional resistance patterns. These updated regimens, such as combinations of bedaquiline, delamanid, linezolid, and other key medicines, are based on evidence from recent clinical studies that showed similar or improved outcomes compared with traditional longer treatments while enhancing patient adherence and tolerability.
Shorter protocols have been incorporated into national programmes in several countries, where they have led to higher completion rates and lower treatment related toxicity. Operational research and policy support, including the WHO’s guidance and support tools, help programme managers introduce these regimens safely into routine care. The move toward condensed treatment courses represents a major opportunity: by reducing the overall duration of therapy, it lessens the physical, financial, and social impacts on patients, lowers the risk of adverse events, and enables health systems to allocate resources more efficiently.
Category-wise Analysis
Drug Class Insights
Bedaquiline is anticipated to dominate, holding approximately 60% share in 2026. Bedaquiline has emerged as a cornerstone in the treatment of multidrug-resistant tuberculosis (MDR-TB) due to its potent activity against resistant strains and inclusion in WHO-recommended all-oral regimens. Unlike traditional second-line drugs, it targets the mycobacterial ATP synthase enzyme, providing a novel mechanism to combat TB bacteria. Its oral administration improves patient adherence compared with injectable therapies, while clinical trials have demonstrated higher cure rates and reduced treatment durations.
In India, bedaquiline is central to treating drug resistant TB through the six month BPaL M regimen (bedaquiline, pretomanid, linezolid, ± moxifloxacin) under the National TB Elimination Programme, benefiting over 1,000 patients in Mumbai by improving adherence and treatment outcomes.
Clofazimine represents the fastest-growing drug class, due to its integration into newer WHO-recommended regimens for MDR-TB and XDR-TB. Originally used for leprosy, it has proven effective against Mycobacterium tuberculosis when combined with other second-line drugs. Its oral administration improves patient adherence compared with injectable alternatives, and it is generally well-tolerated, with manageable side effects. Clofazimine, sold under the brand Lamprene®, was originally developed and manufactured by Novartis Pharmaceuticals Corporation and is also available in generic versions, such as those produced by Macleods Pharmaceuticals Ltd., for use in drug resistant TB treatment regimens.
Disease Type Insights
Active TB is projected to dominate, holding 72% of the market share for 2026, due to patients with active drug resistant disease requiring urgent, prolonged, and often intensive pharmacotherapy to stop transmission and reduce mortality. Active TB drives the highest treatment volume and urgency, which leads to larger drug procurement and usage compared with latent infections that need only preventive therapy and occur asymptomatically. Johnson & Johnson’s anti-TB medication, bedaquiline (Sirturo®), is designed to treat multidrug-resistant TB (MDR TB). It was approved for use when standard first-line TB therapies are ineffective, targeting the urgent treatment needs of patients with active, drug-resistant infections.
Latent TB is the fastest-growing disease type, driven by global preventive therapy initiatives. Individuals with latent TB infection (LTBI) carry the bacteria without showing symptoms but remain at risk of progressing to active TB, particularly in high-burden regions. Governments and health organizations are increasingly focusing on screening, prophylactic treatment, and vaccination programs to curb the future burden of active DR-TB. Sanofi’s rifapentine, sold as Priftin®, is administered once weekly for 12 weeks in combination with isoniazid (the 3HP regimen) to treat latent tuberculosis infection (LTBI) and help prevent its progression to active TB.
Regional Insights
North America Drug-Resistant Tuberculosis Market Trends
North America is projected to dominate approximately 36% of revenue in 2026, shaped by evolving clinical needs, rising public health focus, and enhanced diagnostic and treatment frameworks. Increasing awareness of multidrug resistant (MDR TB) and extensively drug resistant TB (XDR TB) has prompted healthcare systems to adopt more targeted screening and therapeutic strategies. Emphasis on early detection through molecular diagnostics and rapid susceptibility testing is helping clinicians tailor regimens more effectively, reducing treatment delays and improving patient outcomes. This shift toward precision management is supporting demand for advanced diagnostic platforms alongside second and third line therapeutic agents.
Treatment paradigms are shifting with newer oral drugs gaining preference due to better safety profiles and convenience relative to older injectable regimens. The incorporation of novel agents into treatment protocols has sparked interest among clinicians and payers, particularly for populations with limited response to traditional therapies. Public and private healthcare stakeholders are also focusing on patient adherence programs and integrated care models to address the prolonged treatment durations associated with DR TB, as these are essential for achieving sustained success and preventing further resistance.
Policy and funding initiatives at the federal and state levels are reinforcing surveillance efforts and facilitating access to newer therapies, particularly for high risk and underserved communities. Collaboration between academic institutions, healthcare providers, and advocacy groups is strengthening research into next generation anti TB drugs and resistant strain profiling.
Europe Drug-Resistant Tuberculosis Market Trends
Europe's drug resistant tuberculosis (DR TB) remains a significant public health challenge despite steady declines in overall TB incidence over the past decade. The World Health Organization (WHO) European Region continues to report disproportionately high rates of multidrug resistant TB (MDR TB) and pre extensively drug resistant TB compared with global averages, particularly in Eastern European and Central Asian countries, where MDR TB prevalence remains elevated. This sustained burden drives continued demand for advanced diagnostics, rapid drug susceptibility testing, and effective treatment regimens tailored to resistant strains.
A critical trend shaping the market is the emphasis on early and accurate detection of drug resistance. Public health authorities are scaling up access to WHO recommended rapid molecular diagnostic tools to shorten time to diagnosis and initiate appropriate therapy sooner, which can reduce transmission and improve outcomes. There is also growing adoption of shorter, all oral treatment protocols for DR TB, which improve patient adherence and reduce reliance on injectable agents with higher toxicity.
Despite progress, a persistent gap in case detection and follow up undermines efforts to control DR TB. A notable proportion of TB cases in the European Region remain undiagnosed or unreported, contributing both to ongoing transmission and the emergence of resistance due to delayed or incomplete treatment.
Asia Pacific Drug-Resistant Tuberculosis Market Trends
Asia Pacific is likely to be the fastest-growing region, powered by evolving public health priorities, expanding diagnostic capabilities, and enhanced access to second line therapies. A major trend is the region’s increased focus on early detection and diagnosis of DR TB, driven by national TB programs investing in rapid molecular testing and wider screening initiatives. Countries with high TB burdens are scaling up implementation of GeneXpert and other advanced diagnostics, which help identify resistant strains quickly and support timely initiation of appropriate treatment.
An important development is the shift toward shorter, more tolerable treatment regimens. Traditional long duration therapies have had low adherence, contributing to resistance development. In response, healthcare systems are adopting newer drug combinations and repurposed agents that offer improved outcomes and reduced side effects, encouraging better patient compliance.
Public-private partnerships and international collaborations are also influencing market dynamics. Governments are collaborating with global health agencies and pharmaceutical companies to expand the availability of key medications and training for healthcare providers. This collaborative trend strengthens supply chains and ensures more consistent access to essential drugs in both urban and rural settings. Preventive care for latent TB infection is gaining traction as a complementary strategy to active disease management. With greater emphasis on preventive therapy and community outreach, the latent TB segment is expanding, reflecting a strategic shift toward reducing future active DR TB cases.
Competitive Landscape
The global drug-resistant tuberculosis market is highly competitive, driven by the urgent need for effective treatments against multidrug-resistant (MDR) and extensively drug-resistant (XDR) TB strains. Leading pharmaceutical companies such as Sanofi SA, Novartis AG, Endo International Plc, CMP Pharma Inc., STI Pharma LLC, Akorn Incorporated, Lupin, and Johnson & Johnson Services Inc. are at the forefront, focusing on the development of new anti-TB drugs and combination therapies that improve treatment outcomes while minimizing side effects.
Competition in the market is not limited to drug innovation; it extends to efficacy, safety, affordability, and the ability to address resistant TB strains effectively. Companies are actively working on shorter-duration regimens to enhance patient adherence, reduce treatment failure, and limit the emergence of further resistance. Major players are expanding access through public-private partnerships and global health collaborations, particularly in high-burden regions such as Asia, Africa, and Eastern Europe. Investments in research and development for novel compounds, including repurposed antibiotics and new chemical entities, remain central to maintaining a competitive edge.
Key Industry Developments:
- In April 2025, the World Health Organization updated TB guidelines, introducing a six-month all-oral regimen for MDR and RR TB, including pre-XDR cases, and modified nine-month regimens for patients without fluoroquinolone resistance. The changes aim to improve effectiveness, reduce side effects, and enhance patient adherence.
Companies Covered in Drug-Resistant Tuberculosis Market
- Sanofi SA
- Novartis AG
- Endo International Plc
- CMP Pharma Inc.
- STI Pharma LLC
- Akorn Incorporated
- Lupin
- Johnson & Johnson Services Inc.
- Macleods Pharmaceuticals Ltd.
- Pfizer Inc.
- Hikma Pharmaceuticals PLC
Frequently Asked Questions
The global drug-resistant tuberculosis market is projected to reach US$1.5 billion in 2026.
The drug-resistant tuberculosis market is primarily driven by the rising prevalence of multidrug-resistant TB and increasing government efforts for TB elimination.
The drug-resistant tuberculosis market is poised to witness a CAGR of 7.3% from 2026 to 2033.
Key opportunities in the drug-resistant tuberculosis market include the development of shorter treatment regimens, expansion in high-burden emerging markets, and growth in latent TB preventive therapy.
Key players in the drug-resistant tuberculosis market include Pfizer Inc., Johnson & Johnson Services Inc., Novartis AG, Sanofi SA, and Lupin.







